Two-Patch Repair of Rastelli’s Type-A Complete Atrioventricular Septal Defect, Pulmonary Valvotomy and Relief of Right Ventricular Outflow Tract Obstruction under Mild Hypothermic Extracorporeal Circulation and Cardioplegic Arrest: A Video Presentation
Ujjwal K. Chowdhury, Niwin George, Sukhjeet Singh, Lakshmi Kumari Sankhyan, Suryalok Angadi, Chaitanya Chittimuri, Doniparthi Pradeep, Saidivya Yadavalli
Cardiothoracic Sciences Centre, All India Institute of Medical Sciences, India
Abstract
Video File: www.jsurgery.com/articles/video/JSST-20-1111.mp4
The association of right ventricular outflow tract obstruction with complete atrioventricular septal defect is rare and poses surgical challenge at the time of intracardiac repair. We report here-in a 30-months-old female child diagnosed with severe right ventricular outflow tract obstruction, complete atrioventricular septal defect and severe left atrioventricular valvar regurgitation undergoing successful pulmonary valvotomy, resection of the right ventricular outflow tract with reconstruction of the complete atrioventricular septal defect by two-patch technique. The pulmonary valve ring was sutured directly. Postoperative period was uneventful.
Introduction
The atrioventricular septal defects encompass a spectrum of cardiac malformations unified by a deficiency of the atrioventricular membranous and muscular septa1-3. The deficiency of the atrioventricular septum results in an ostium primum atrial septal defect and a scooped-out area in the inlet (basal) portion of the ventricular septum1-3. Abnormal differentiation and remodelling of the cushion mesenchyme into valvulo septal tissue results in a deficiency or absence of the atrioventricular septum4.
At the extreme end of the spectrum lies the complete atrioventricular septal defect characterized by large deficiencies of the atrial and ventricular septa, and a common atrioventricular valvar orifice1-3. Anatomically, all types of atrioventricular septal defects share the following features. These includes: i) absence of wedging of the aortic annulus due to a common atrioventricular valvar ring causing an elongated left ventricular outflow tract and a “goose neck” appearance; ii) lengthened outlet septum-to-ventricular apex ratio; iii) apical displacement of the attachments of the atrioventricular valves to the ventricular crest; and iv) posterior and inferior displacement of the coronary sinus resulting in inferior displacement of the atrioventricular node and elongation of the common atrioventricular bundle1-3,5-7.
Published literature documents a number of morphological factors that complicate the “usual” form of atrioventricular septal defect. These include: i) double outlet left or right atrium8; ii) double orifice left atrioventricular valve9; iii) parachute deformity of the left atrioventricular valve10; iv) variable degrees of underdevelopment of either the right or left ventricle (prevalence of 10%)11,12; and v) variable degrees of left ventricular outflow tract obstruction10,13. Right ventricular outflow tract obstruction complicating complete atrioventricular septal defect has rarely been reported.
Based on the variable anatomical relationship between the superior bridging leaflet with its chordal attachments and the underlying ventricular spetum, Rastelli from Mayo Clinic classified complete atrioventricular septal defect into three types of severity14. In type A defect (69%), the common superior bridging leaflet is committed almost entirely to the left ventricle, and its commissure with the anterior tricuspid leaflet lies along the right anterosuperior rim of the ventricular septum. In the rare type B (9%), the superior bridging leaflet is larger and overhangs the ventricular septum. An anomalous papillary muscle attaches from the right side of the ventricular septum to the left side of the superior bridging leaflet, thereby creating a free interventricular communication. In type C (22%) defects, the superior bridging leaflet is larger, “free-floating”, overhangs the ventricular septum more than in type A or type B without chordal attachment to the ventricular septal crest. The anterior tricuspid leaflet is generally small, supported by the medial papillary muscle14-16.
Patients with complete atrioventricular septal defect without associated cardiac anomalies presenting with congestive heart failure should be operated between 3 and 6 months of age to prevent progressive pulmonary arterial hypertension, congestive heart failure and Eisenmenger’s syndrome17. The principles of surgical repair of complete atrioventricular septal defect include closure of the atrial septal defect, closure of the ventricular septal defect, creation of non-obstructive competent atrioventricular valves, and avoidance of damage to the atrioventricular node and bundle of His15,17,18.
Literature documents three different surgical techniques for repair of complete atrioventricular septal defects. The techniques include: i) the single-patch technique, as described in 1962 by Maloney and Gerbode18,19; ii) the two-patch technique as described by George Trusler in 197620; and iii) the modified single-patch technique of Graham Nunn in 199521,22.
Classic single-patch technique
Although a pericardial, polytetrafluoroethylene or Dacron patch have been used as the patch material, use of pericardium is associated with pericardial patch aneurysm at the ventricular level and the Dacron patch carries the risk of postoperative hemolysis18,19,23.
Two-patch technique
In this technique, the ventricular septal defect is closed using either a Dacron or polytetrafluoroethylene patch and the atrial septal defect is closed using a separate pericardial patch20.
Modified single-patch technique
In this technique, a series of pledget supported 5-0 polypropylene sutures are placed on the right-side of the interventricular septum. These sutures are then passed sequentially through the middle of the superior and inferior bridging leaflet at a site predetermined to separate the mitral and tricuspid valves. Subsequently, these sutures are then placed through the edge of the autologous pericardial patch21,22.
The operative mortality reported in the published literature varies between 3% and 15%17,24,25. The reoperation rates for left atrioventricular valve regurgitation varies between 6% and 9% in different institutions17,24,25.
In 1995, Backer and associates performed a meta-analysis of results among 794 patients undergoing operation by two-patch technique from five institutions7. The operative mortality and the incidence of reoperation for mitral regurgitation were 7% and 8% respectively7. Graham R Nunn and Nicholson reported their results using modified single-patch technique among 72 consecutive patients. The operatively mortality was 2.8%. Postoperative mild and moderate left atrioventricular valve regurgitation was present in 29% and 5% of patients respectively21,22.
We report here-in the surgical reconstruction of the complete atrioventricular septal defect using two-patch technique, pulmonary valvotomy, and resection of the right ventricular outflow tract obstruction. A 30-months old female child presented with poor weight gain, recurrent episodes of chest infection and cyanosis since birth. Clinically, she had cardiomegaly, left parasternal leave, and systolic ejection murmur of grade IV/VI intensity over the 2nd and 3rd left intercostal spaces. Oxygen saturation at rest was 84%-85% as determined by pulse oximetry. Two-dimensional color Doppler echocardiography established the diagnosis of bilateral superior venae cavae, complete atrioventricular septal defect, severe left atrioventricular valvar regurgitation, and severe right ventricular outflow tract obstruction with peak systolic gradient of 110 mmHg across the right ventricular outflow tract. The obstruction was at the level of pulmonary valve and right ventricular outflow tract.
The child underwent successful pulmonary valvotomy, resection of right ventricular outflow tract with reconstruction of complete atrioventricular septal defect by two-patch technique. The pulmonary valve ring was directly sutured.
Although our institution is a tertiary level center, the socioeconomic profile of the patients and lack of health insurance benefit led to delayed referral and surgery.
Surgical Techniques
Patient position
The patient was placed in the supine position.
Intraoperative transesophageal echocardiography
Intraoperative transesophageal echocardiography was performed using a Philips echocardiographic machine to assess the morphology of the atrioventricular valves, size and location of the ventricular septal defects, severity of left atrioventricular valvar regurgitation, degree and level of right ventricular outflow tract obstruction, the sizes of both ventricles and ventricular function (Philips iE 33 echo machine; Philips X7-2T probe, Bothell, WA, USA).
The incision
Following median sternotomy, the thymus was subtotally excised taking care not to expose the brachiocephalic vein. The pericardium was incised on the left side using scissors. We did not use cautery to avoid inadvertent cautery-induced ventricular fibrillation. Three 4-0 silk stay sutures were placed on the incised pericardial edge thus creating a right sided pericardial flap.
Surgical planning
The operation was performed under moderately hypothermic cardiopulmonary bypass with an aortic infusion cannula, angled venous cannulae into both superior venae-cavae and a straight venous cannula into the inferior vena cava. St. Thomas (II) based cold hyperkalemic blood cardioplegia (1:4) and topical ice cooling was used for myocardial preservation.
The persistent ductus arteriosus was clipped using a Hemaclip (Johnson and Johnson Ltd., Ethicon, LLC, San Lorenzo, USA) after pulling down the superior surface of the pulmonary artery at the commencement of cardiopulmonary bypass as described by Dwight McGoon. The pump flow was temporarily lowered at the time of interruption of the ductus arteriosus.
Pulmonary arteriotomy, right atriotomy, insertion of a left ventricular vent
The main pulmonary artery was opened in between stay sutures prior to cross-clamping the aorta to avoid distortion of the main pulmonary artery. The right atrium was opened longitudinally 1 cm posterior to the right atrioventricular groove. Four stay sutures were placed on either side of the right atrial edges. The left ventricle was vented through the atrial septal defect using a DLP vent (Medtronic Inc., Minneapolis, MN).
Relief of right ventricular outflow tract obstruction
The pulmonary valve was tricuspid and stenosed. The stenosed pulmonary valve was incised at the level of pulmonary valve commissures. The obstructing muscle of the right ventricular outflow tract was excised (parietal insertion of the infundibular septum) via combined transpulmonary and transtricuspid approach. Adequacy of right ventricular outflow tract resection was checked by visual assessment and insertion of an appropriately sized Hegars dilator.
Assessment of the atrioventricular valves
The anatomical details of the malformation were examined. The atrial septal defect was of ostium primum type with the posteriorly positioned coronary sinus and margin of the atrioventricular septal defect delineating its boundaries.
The atrioventricular valve was distended by injecting cold saline into the ventricular cavities. This enabled us to delineate the optimal coaptation point of the left superior and inferior bridging leaflets and identify any surgical issues that would require attention during reconstruction. There was a prominent cleft between the left and right superior birding leaflets of Rastelli’s type A variety and a large unrestrictive ventricular septal defect with scooped out ventricular septum.
Dacron patch closure of the ventricular septal defect
The exposure of the ventricular septal defect was facilitated by use of two elastomer vascular loops. It is imperative to measure the length and height of the ventricular septal defect. The distance between the crest of the ventricular septum and the point of coaptation of the left superior and inferior bridging leaflets was considered as the height of the ventricular septal defect.
In order to minimize postoperative left atrioventricular valve regurgitation, it is important to maintain a smaller height of the ventricular septal patch to prevent/ minimize iatrogenic postoperative left atrioventricular valvar regurgitation, which is classically “D-shaped”. A series of pledget supported interrupted 5-0 polypropylene sutures (Johnson and Johnson Ltd., Ethicon, LLC, San Lorenzo, USA) were placed 0.5 cm away from the ventricular septal crest.
At the superior and inferior corners of the ventricular septal defect, extreme precautions were taken to prevent residual ventricular septal defect. In both superior and inferior corners, the sutures were brought through the annulus and the substance of the superior and inferior bridging leaflet to prevent residual ventricular septal defect. The ventricular septal defect was closed using a “D-shaped” Dacron polyster patch (Bard® Savage® filamentous knitted polyester fabric, Bard Peripheral Vascular Inc., Tempe, AZ, USA), slightly smaller than the size of the ventricular septal defect.
Reconstruction of the left atrioventricular valve
The septal commissure was reconstructed using Alain Carpentier’s technique. A stay suture of 6-0 polypropylene was placed at the free leaflet margin opposing the atrial edge of the coaptation border. The septal commissural cleft was repaired using multiple interrupted, non-pledgeted 6-0 polypropylene sutures, taking precautions to take the bites through the atrial edge and not the ventricular edge, thus ensuring perfect competence15.
The newly constructed left atrioventricular valve opening was sized and matched with the indexed mitral valve orifice appropriate for the patient, thus avoiding iatrogenic mitral stenosis15. Cold saline was injected into the left ventricular cavity to ensure competence of the left atrioventricular valve and any leaking commissures, if present.
Pericardial patch closure of the atrial septal defect
A series of interrupted 5-0 polypropylene sutures were used between top of the ventricular septal patch and pericardial margin thus sandwitching the left atrioventricular valve. Extreme precautions were exercised at the superior and inferior corners of the defect, ensuring that the ventricular septal patch was brought into the corners of the superior and inferior bridging leaflets, thus preventing residual defects.
Subsequently, the pericardial patch was sutured to the edge of the ostium primum defect. In the area of the bundle of His, the sutures were passed through the superior edge of the coronary sinus, thus diverting the coronary sinus to the left side. Before completing the suture line, the left side of the heart was filled with cold saline and gentle ventilation was resumed to evacuate residual air, if any.
Assessment of the right atrioventricular valve
Cold saline was injected into the right ventricle to ensure competence of the right atrioventricular valve. The right atrioventricular valve was competent. No additional reconstructive procedures were required.
Closure of the right atriotomy and weaning off bypass
The right atrium is closed in two layers using 5-0 polypropylene suture. The pulmonary valve ring was closed directly in two layers using 5-0 polypropylene sutures. The patient was weaned off cardiopulmonary bypass with stable hemodynamics. The aortic cross clamp and cardiopulmonary bypass times were 62 minutes and 84 minutes respectively.
The postoperative peak systolic right ventricle-to-left ventricle pressure ratio was 0.5. Intraoperative transesophageal echocardiography did not demonstrate any residual ventricular septal defect. There was no left or right atrioventricular valvar regurgitation and no significant residual right ventricular outflow tract gradient.
Short- and Long-term Results
Postoperatively, the child was in normal sinus rhythm and recovery was uneventful. At 24th month follow-up the child was asymptomatic, no clinical evidence of cardiac failure, with Ross’s clinical score of 2. Two-dimensional color flow Doppler echocardiography revealed normal biventricular function with minimal atrioventricular valvar regurgitation, intact atrial and ventricular septation with mild mitral and tricuspid regurgitation. The peak systolic right ventricular outflow tract gradient was 27 mmHg with minimal pulmonary regurgitation (Figures 1A-1G).
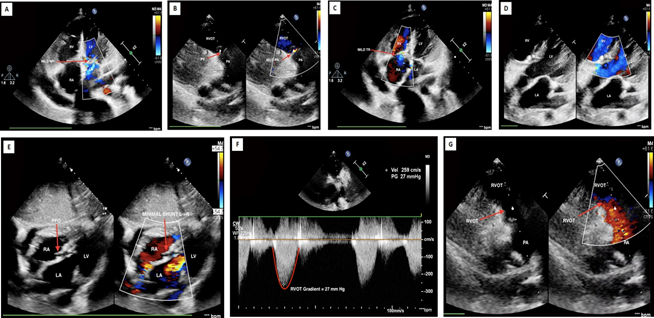
Figures 1A-1G: Series of post repair intraoperative transesophageal two-dimensional color flow Doppler echocardiography showing normal biventricular function with minimal atrioventricular valvar regurgitation, intact atrial and ventricular septation with mild mitral and tricuspid regurgitation. The peak systolic right ventricular outflow tract gradient was 27 mmHg with minimal pulmonary regurgitation.
Study Limitations
Because of limited availability of slots for magnetic resonance scanning in the institute and the small age of the child which would have necessitated heavy sedation or general anaesthesia on account of longer scan duration in magnetic resonance imaging, magnetic resonance angiography could not have be performed.
Conclusions
Surgical repair of severe right ventricular outflow tract obstruction and left atrioventricular valve regurgitation should be undertaken in early childhood to prevent further anatomical deterioration/deformation of the left atrioventricular valve and to preserve ventricular function. Adequate resection of the right ventricular outflow tract without creating pulmonary regurgitation is of paramount importance to prevent right ventricular dysfunction, thus providing good long-term outcome.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of the article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
References
- Anderson RH, Zuberbuhler JR, Penkoske PA, et al. Of clefts, commissures and things. J Thorac Cardiovasc Surg. 1985; 90: 605-610.
- Bharati S, Lev M, McAllister HA Jr, et al. Surgical anatomy of the atrioventricular valve in the intermediate type of common atrioventricular orifice. J Thorac Cardiovasc Surg. 1980; 79: 884.
- Jacobs JP, Burke RP, Quintessenza JA, et al. Congenital Heart Surgery Nomenclature and Database Project: Atrioventricular canal defect. Ann Thorac Surg. 2000; 69 (suppl): 36.
- Eisenberg LM, Markwald RR. Molecular regulation of atrioventricular valvuloseptal morphogenesis. Circ Res. 1995; 77: 1-6.
- Anderson RH, McCartney FJ, Shinebourne EA, et al. Pediatric Cardiology, Churchill Livingstone, Edinburgh. 1987; 571-613.
- Anderson R, Baker E, Ho S, et al. The morphology and diagnosis of atrioventricular septal defects. Cardiol Young. 1991; 1: 291.
- Backer CL, Mavroudis C, Alboliras IT, et al. Repair of complete atrioventricular canal defects: results with the two-patch technique. Ann Thorac Surg. 1995; 60: 530.
- Suzuki Y, Hamada Y, Miura M, et al. Double-outlet left atrium with intact ventricular septum. Ann Thorac Surg. 1988; 45: 332-334.
- Ilbawi MN, Idriss FS, DeLeon SY, et al. Unusual mitral valve abnormalities complicating surgical repair of endocardial cushion defect. J Thorac Cardiovasc Surg. 1983; 85: 697-704.
- De Biase L, Di Ciommo V, Ballerini L, et al. Prevalence of left-sided obstructive lesions in patients with atrioventricular canal without Down syndrome. J Thorac Cardiovasc Surg. 1986; 91: 567-472.
- Freedom RM, Bini M, Rowe RD. Endocardial cushion defect and significant hypoplasia of the left ventricle: A distinct clinical and pathological entity. Eur J Cardiol. 1978; 7: 263-281.
- Corno A, Marino B, Catena G, et al. Atrioventricular septal defects with severe left ventricular hypoplasia. J Thorac Cardiovasc Surg. 1988; 96: 249-272.
- Chang CI, Becker AE. Surgical anatomy of left ventricular outflow tract obstruction in complete atrioventricular septal defect. A concept for operative repair. J Thorac Cardiovasc Surg. 1987; 94: 897-903.
- Rastelli G, Kirklin JW, Titus JL. Anatomic observations on complete form of persistent common atrioventricular canal with special reference to atrioventricular valves. Mayo Clinic Proc. 1966; 41: 296.
- Carpentier A. Surgical anatomy and management of the mitral component of the atrioventricular canal defects. In Anderson RH, Shinebourne EA, editors. Pediatric Cardiology 1979; 466-490. London: Churchill Livingstone.
- Torfs CP, Christianson RE. Anomalies in Down syndrome individuals in a large population-based registry. Am J Med Genet. 1998; 77: 431-438.
- Hanley FL, Fenton KN, Jonas RA, et al. Surgical repair of complete atrioventricular canal defects in infancy. Twenty-year trends. J Thorac Cardiovasc Surg. 1993; 106: 387.
- Maloney IV Jr, Marable SA, Mulder DC. The surgical treatment of common atrioventricular canal. J Thorac Cardiovasc Surg. 1962; 43: 84.
- Gerbode F. Surgical repair of endocardial cushion defect. Ann Chir Thorac Cardiovasc. 1962; 1: 753.
- Trusler GA, Mills NL, Ochsner JL, King TD. Discussion of Correction of type C complete atrioventricular canal. Surgical considerations. J Thorac Cardiovasc Surg. 1976; 71: 20.
- Nunn G. AV canal repair: without VSD patch. Congenital Heart Disease Symposium, Eighty-first Annual Meeting. The American Association for Thoracic Surgery, San Diego, May 6, 2001.
- Nicholson IA, Nunn GR, Sholler GE, et al. Simplified single-patch technique for the repair of atrioventricular septal defect. J Thorac Cardiovasc Surg. 1999; 118: 642.
- Burkhart, HM, Moody SA, Ensing GH, et al. Ventricular septal aneurysm after atrioventricular septal repair with pericardium. Ann Thorac Surg. 1996; 61: 1838.
- Capouya ER, Laks H, Drinkwater DC Jr, et al. Management of the left atrioventricular valve in the repair of complete atrioventricular septal defects. J Thorac Cardiovase Surg. 1992; 104: 196.
- Merrill WH, Hammon JW Jr, Graham TP Jr, et al. Complete repair of atrioventricular septal defect. Ann Thorac Surg. 1991; 52: 29.
