Arthroscopy in Rheumatology: A Reminiscence
Robert W. Ike1*, William J. Arnold2, Kenneth C. Kalunian3
1 Department of Internal Medicine, Division of Rheumatology, University of Michigan Health System
2 Rheumatologist, Orthopaedics and Rheumatology of the North Shore, Wilmette, IIlinois
3 Department of Medicine, Division of Rheumatology, Allergy and Immunology,University of California at San Diego
Abstract
Rheumatology is defined by the many medical conditions that can affect joints, but rheumatologists have only been intermittently interested in looking into them. Rheumatologists were involved in the early development of arthroscopy, and most of the surgeons looking into joints were examining the sorts of conditions today’s rheumatologists would handle.
Physicians and surgeons first began adapting endoscopes to peer into joints mainly to assess the sort of synovial conditions that would concern today’s rheumatologists. Rheumatologists were among the pre-World War II pioneers developing and documenting arthroscopy. Post-war father of modern arthroscopy Watanabe found rheumatologists among his early students, who took back the technique to their home countries, teaching others. Rheumatologists described and analyzed the intraarticular features of their common diseases in the 60s and 70s. Improvements in instrumentation and dogged efforts by a few orthopedists made several common joint surgical procedures feasible under arthroscopic guidance. A groundswell of interest from academic rheumatologists adapting arthroscopy grew considerably in the 90s with the development of “needle scopes” that could be used in an office setting. Rheumatologists had a hand in their downfall, conducting the first prospective trial questioning the efficacy of arthroscopic debridement in OA and developing biological compounds that greatly reduced the call for any resective intervention in inflammatory arthropathies. The arthroscope has proven an excellent tool for viewing and sampling synovium and continues to serve this purpose at several international research centers. While cartilage is imaged mainly by MRI now, some OA features – such as a high prevalence of visible calcinosis – beg further arthroscopy-directed investigation.
A new generation of “needle scopes” with far superior optics awaits for future investigators, should they develop interest.
Beginnings
Once Edison’s incandescent light made illumination of various body cavities practical, physicians and surgeons began to adapt and modify cystoscopes and laparoscopes to examine joints1. After first showing it was even feasible, their focus was trained mainly on chronic inflammatory conditions of the sort that might concern today’s rheumatologists, particularly then prevalent tuberculosis. Nordentoft, a Dane, is credited as being the first arthroscopist, but after showing these inspections to be feasible, he developed no clinical applications2. Takagi took a very large instrument by today’s standards to examine tuberculous knees and began the line of Japanese arthroscopists that would lead to Watanabe and modern arthroscopy1. Both Bircher, a Swiss, and Kreuscher, a Chicagoan focused on internal derangements of the knee, but both abandoned the technique out of frustration for lack of acceptance1. Burnam was one of three orthopedists at NYU’s Hospital for Joint Diseases working on arthroscopy, traveling to Dresden for part of his anatomic studies3.
As he was publishing his findings, colleagues Finkelstein and Mayer had used a scope they’d had constructed to examine patients with unexplained synovial disease, mainly suspected TB4. Later they combined forces and also looked at internal derangements5.
North Dakota’s Geist examined cadaver knees with a modified otoscope and commented “The performance of arthroscopy in the living ought to be no more stress to the patient than a simple aspiration, provided local anesthesia is employed”, a standard to which we office arthroscopists still strive to attain6. Rheumatologist Ernst Vaubel of Germany7, joined by fellow rheumatologist Sommer8, pursued arthroscopy, and documented his systemic review of arthritic conditions in a book, the first book on arthroscopy; his practice did not survive the war, but his concepts and equipment designs were rediscovered in the mid-50s by 2 French orthopedists9,10 whose appropriation of his designs influenced Wolf as they developed arthroscopes. The fragility of various scopes and difficulty of the procedure prevented wider use. World War II stopped all experimentation in arthroscopy.
After the War
Makei Watanabe reentered civilian life, securing a post at the post office hospital in Tokyo11. He had been a student of Takagi and inherited his 14 unsuccessful pre-war arthroscope designs. He noted that many of the patients with arthritis he'd arthroscoped felt better after what was essentially a diagnostic procedure. He devised a technique – “articular pumping” – by which the joint could be lavaged without an arthroscope and reported positive results12. By the mid-50s, he had taken color photos and made movies from arthroscopy, and introduced the concept of triangulation, whereby instrumentation inserted from a different site in the knee can be seen by the arthroscope. He presented his movie throughout Europe and the U.S. in 1957, but despite being well received, it did not engender a groundswell of enthusiasm for arthroscopy. He continued to work on an arthroscope, and his 21st design in 1959 seemed workable and went into production. Illumination came from an incandescent light at the end of the scope, leading to occasional shocks to the patient during the procedure (Figure 1). He published an atlas of his findings in 195713 and updated the painted images with color photographs in 196914. He was the first to attempt resective procedures under arthroscopic guidance, removing a giant cell tumor in 1955, a loose body in 1961, and part of a meniscus in 1962. His student Takeda explored correlations between macroscopic and histologic characteristics of synovitis15. He invented the first needle scope in 1970, the 2.0 mm diameter Selfoc arthroscope, which made arthroscopy of small joints feasible and was employed extensively by Johnson, mainly for the knee, in his early years. He was not well known, even in his native Japan, but visitors began to seek him out. Watanabe trained rheumatologists from South America and Europe, including Robes-Gil and Katona of Mexico16, Lombas Garcia and Griego Gonzalez of Cuba17, Marques and Barceló of Spain18, Andersen of Denmark19, and Dorfman of France20. Each student subsequently trained both orthopedists and rheumatologists from home country and abroad with some publishing textbooks21,22.
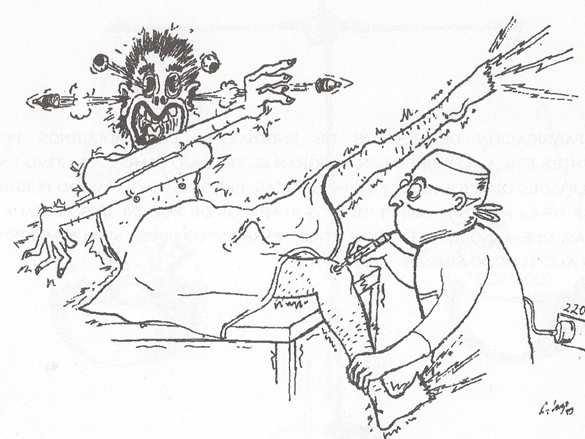
Figure 1: Patients undergoing arthroscopy with the #21 scope sometimes experienced shocks from the incandescent bulb at the tip of the scope. Students of Watanabe who took the #21 home had the same experience. This extended well into the 70s till fiberoptics took hold. From Checa A and Lazo A. Artoscopia. Pasajies y memorias. Havana: CIMEQ press hospital, 1996, with permission.
Rheumatologists performed most of the arthroscopies in Cuba and France into the 21st century.
Watanabe’s most influential visitor was the late Canadian orthopedist Bob Jackson who returned to Toronto after the ’64 Olympics and worked to master the #21 arthroscope while adapting common knee surgeries to performance under arthroscopy23. In ’69, Jackson spent time with Miami rheumatologist Roy Altman, who then began performing arthroscopy independently under local anesthesia, serving as the only arthroscopist in south Florida for a decade while describing the intraarticular pathology in pseudogout24 and offering guidelines on the use of arthroscopy in rheumatology25. He helped form the International Arthroscopy Association in 1974 and was present at the beginnings of the Arthroscopy Association of North America (AANA).
Several British Rheumatologists started to publish their findings on synovial diseases26-28. Yates wrote the first chapter touching on arthroscopy in Copeman’s Textbook of Rheumatology29, repeated in all subsequent iterations. The first British orthopedist to publish on arthroscopy, Dandy, did not do so till ‘7530.
Watanabe applied fiber light to the arthroscope in 1967, replacing the bulb.
The adaptation and miniaturization of video to arthroscopy brought safety, sharing, and the possibility of a permanent record for each procedure31. Johnson’s motorized shaver made extensive tissue resection feasible32. All these developments made arthroscopy a far less daunting procedure, not only for orthopedists but for rheumatologists as well.
The 80s
In 1981, Bill Arnold of Chicago met orthopedist Dave Stulberg of Northwestern, who was employing arthroscopic synovectomy to good effect in patients with RA33. The two set up a cooperative arthroscopy unit to explore further the nuances of arthroscopic interventions. (Figure 2)
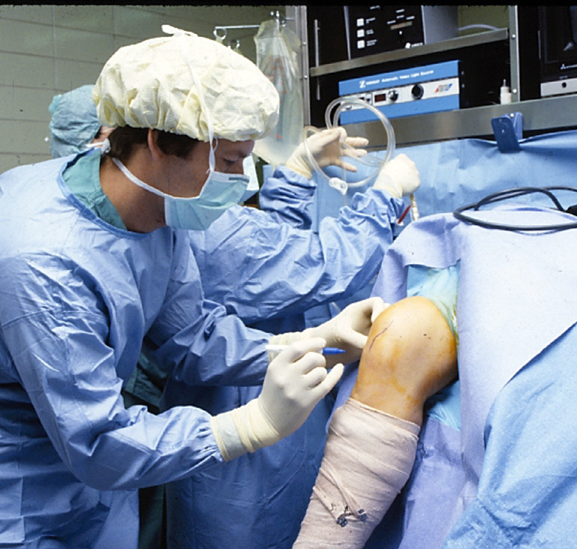
Figure 2: Arnold performing outpatient arthroscopy. Augustana Hospital, Chicago c1983.
Rheumatologists performed most of the arthroscopies in Cuba and France into the 21st century.
Arnold’s mentor during his fellowship at Duke Bill Kelley became Chief of Medicine at the University of Michigan in ’79 and explored expansion into modern technologies for all the medicine subspecialties. Impressed with his former fellow’s progress, he sought to identify a trainee who could go learn from Arnold and then carry out arthroscopy in an academic setting. 6’8” Bob Ike was chosen, in part because Kelley knew that orthopedists respected size. After some early struggles, arthroscopy was going on at U of M by ’87. That same year, Kelley was President of the American Rheumatism Association (now American College of Rheumatology). In his presidential address in June, he lamented that so few medical students and residents were interested in rheumatology. Among the several approaches he proposed taking to enhance interest in rheumatology, he stated, “I believe we need to expand the specialty of rheumatology to cover some of the peripheral areas which now are largely ignored and sometimes poorly handled. This would include …the use of certain technical procedures which are appropriate to our specialty” (my emphasis)34.
From Sweden, Lindblad and Hedfors gave a hint as to where arthroscopic research into synovium might go, describing correlation of macroscopic and microscopic features of synovitis35. Combs of Mayo Clinic described the differential diagnosis of synovial proliferative disorders36.
The ARA Board of Directors in June ’86 approved guidelines for the performance of arthroscopy by rheumatologists37. AANA guidelines were not published till ’9338, but did include a path for non-orthopedists to perform non-operative arthroscopy and were published in every issue of Arthroscopy until January ’19.
Bevra Hahn of UCLA sent Ken Kalunian in ’88 to study with Bill Arnold, where their cases included arthroscopic synovectomies. Arnold and Stulberg conducted trials comparing joint debridement to closed lavage in knee OA, finding no appreciable difference in outcomes39. An accompanying editorial “why a rheumatologist should be interested in arthroscopy.” emphasized how arthroscopy could help increase understanding of the pathoanatomic anatomy rheumatologists confront40. Counters to the editorial cited mainly concerns about malpractice coverage41.
French rheumatologists Combe et al. described the effectiveness of arthroscopic synovectomy in patients who had failed radiation synovectomy42. In an accompanying commentary, Arnold and Kalunian described their experiences with arthroscopic synovectomy in refractory rheumatoid knees37.
The 90s
The appearance in 1990 of fiberoptic scopes that were 1.7 mm in diameter rather than the 4.0 mm glass lens scope made possible the performance of arthroscopy in an office or procedure room setting rather than the operating room. Leaders in orthopedics and rheumatology recognized the potential advantages of moving arthroscopy into an office setting. Arnold, a rheumatologist, cited the potential to evaluate the contribution of intraarticular pathologies to various clinical scenarios while providing a means to assess synovium43. Halbrecht, the orthopedist, described 21 patients in which assessment by office needle arthroscopy provided information equivalent to MRI, proposing the technique as an alternative44. Reece and Emery, speaking from a center that would become a leader in applications of arthroscopy to the investigation of the synovium over the next decade, emphasized what might be learned by evaluating early synovitis, including smaller joints now accessible by needle scopes45.
The ”needle scope” permitted characterization of intraarticular pathology sufficient for the rheumatologist’s need46 although it missed some lateral compartment pathology and some orthopedists found it missed some other structural pathology47,48. With advances in instrumentation now permitting arthroscopy to be performed in a procedure room or office, interest among rheumatologists surged in the early 1990s. Instructional courses sponsored by the American College of Rheumatology (ACR) and commercial concerns were highly popular.
The ACR convened committees to address several issues in arthroscopy and arthroscopy study group meetings became a regular part of every national ACR yearly meeting. A 1992 survey of the 14 NIH Multipurpose Arthritis Centers found 6 where a rheumatologist was already performing arthroscopy and 7 more where a faculty member was either already in training or planning to do so49. Chapters focusing on arthroscopy began appearing in major rheumatology textbooks by the mid-90s. Kalunian’s chapter50 ran through the end of Koopman and Moreland’s run, while Ike’s chapter51, with different authors, continues in Hochberg.
The use of arthroscopy spread more widely among European rheumatologists than in the U.S. Reports from Belgium52 and Down Under53 highlighted the feasibility, tolerance, and safety of the procedure in large groups of patients, done primarily for research purposes. Michalska described a clinical situation in Chicago54. Wei, an aggressive American rheumatologist, described the resective procedures he was able to accomplish in an office setting but emphasized the procedure was still an art form, requiring a gentle touch55.
The arthroscopic view of patients we had been approaching blindly for decades led to some revelations (Figure 3). Intraarticular abnormalities in OA strongly correlated with cartilage damage56, as was shown by subsequent MRI studies57. Cartilage pathology seen at arthroscopy could be accurately predicted by specific physical exam maneuvers58. Reece and colleagues found that that the tortuous vascular pattern of patients with psoriatic arthritis and the spondyloarthropathies was much different than the linear vessels seen in RA59. Cañate extended these observations to note that such synovial vascular tortuosity was also seen in OA, although to a much lesser extent60. Moll and colleagues later described distinct macroscopic features of Behçets61. Ike and colleagues found that of patients with knee OA undergoing arthroscopic lavage, 39% had macroscopic features of calcinosis62. Recently, Wei arthroscoped 57 patients with OA but no chondrocalcinosis and found visible calcific deposits in 38 (67%), identifying CPPD in the synovial fluid of 2463.
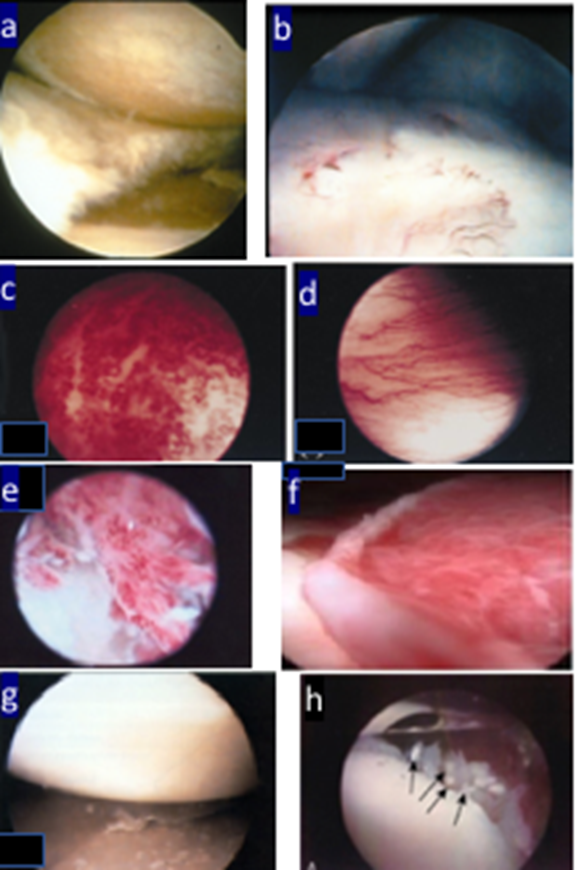
Figure 3: Arthroscopic views of intraarticular pathology in various rheumatic conditions. (a) crushed meniscus between two surfaces of nearly bare bone (thanks to WJ Arnold). (b) early pannus at bone cartilage junction, just as described by Robes-Gil 16 (thanks to WJ Arnold). Second row: examples of the “squiggly vessels (c) in synovium of psoriatic arthritis, reactive arthritis, and the spondyloarthropathies compared to “straight vessels” of RA (d) (reference 59, with permission). In the next row on the left (e) “squiggly vessels in an OA knee (reference 60, with permission). (f) a patient with Behçet’s showing extensive erythematous synovium without villi or a distinct vascular pattern (reference 61, with permission). To the lower left is (g) a patient with gout showing deposits on cartilage (reference 51, with permission). (h) calcific deposits in an OA knee (reference 62, with permission).
Ayral and colleagues devised a system whereby articular cartilage lesions seen at arthroscopy might be assigned a semi-quantitative score (“chondroscopy”), providing a means by which interventions directed at osteoarthritis and cartilage might be assessed serially64. When this system, and several others, were assessed for interobserver and intraobserver variability, it became apparent the system would work only with a solitary assessor65. Ayral also emphasized the importance of the arthroscope, with the quality of views from the popular fiberoptic needle scopes not matching what was seen through the conventional glass lens scopes66. The first hint that direct visualization might not be necessary to assess cartilage came when Brodenick and colleagues showed that fast-spin echo MRI assessed cartilage lesions as well as did arthroscopy67.
While few rheumatologists took up mechanical resective procedures, some explored other means of tissue modification. Laing et al. demonstrated that synovitis in a patient pretreated with psoralens could be modified by the fiberoptic arthroscopic application of ultraviolent light energy68. Wei, who had been doing tissue resections, found the holmium YAG laser to be an acceptable and more convenient substitute for the obliteration of pathologic soft and hard tissue in 34 cases69, with speedier and less painful recovery compared to conventionally treated patients70.
The prevalence of symptomatic knee arthritis coupled with many possible costly arthroscopic interventions inspired research on outcomes from these procedures. Prospective trials of arthroscopic debridement in knee OA could show no benefit over placebo71,72. Lavage of the joint with large volumes of saline – which occurs during every arthroscopy regardless of whether any surgery is performed - also was shown to add nothing to placebo in knee OA73. However, Kalunian and colleagues demonstrated that OA patients undergoing arthroscopic lavage did enjoy better outcomes after a larger volume lavage than a small volume one, and those with macroscopic calcinosis did best74. These and other data essentially shut down all rheumatologic arthroscopy for OA, they have not yet completely permeated orthopedic practice75. The gratifying responses to biologics in RA made it far less greatly that any inflamed knee would become so refractory to therapy to be considered for surgery, greatly reducing the number of knees that might be considered for synovectomy - a difficult procedure to support if few other arthroscopic procedures are being done.
Yousswel and colleagues could find no differences in synovial tissue obtained by blind biopsy with that from arthroscopy75. Yet, interest in arthroscopy as a means to evaluate early synovitis remained keen76. Practitioners at 3 centers reported complication rates, all minimal and minor. Hemarthroses, complicating 3.5% of cases at one report77, were not seen in the other center which did not do the same array of resective procedures78. There were no infections.
The 21st century
The new century found the use of arthroscopy by rheumatologists fading. Ike did his last case in May 2001, Arnold in 2004, and Kalunian concluded his last study in ’0780. In a few international centers, arthroscopy became integrated into the necessary research tools for studying the synovium. A 2002 survey of arthroscopy performed by rheumatologists found 36 centers where arthroscopy was still being performed, 10 in the US; only 15% were doing resective interventions, with research/clinical breakdown at 50/5081. Reports of arthroscopy by rheumatologists emerged from exotic outposts like Uruguay82 and Bulgaria83. Reece produced a detailed treatise on the use of arthroscopy as a tool to investigate synovial disorders, including instructions on tissue handling84. Clinical applications persisted85,86, and new interest emerged from the subcontinent87. The foray into small joints became a reality88. Therapeutic effects of arthroscopy would seem limited, absent a surgical component, but several studies have shown that the joint lavage accompanying the procedure, discredited in osteoarthritis, can have positive effects in inflammatory arthropathies89, particularly if delivered in large volume90 and followed by a corticosteroid injection91.
With techniques for analysis of synovial tissue becoming far more powerful, the biopsy of synovium not only for classic diagnosis but to provide a step-change towards personalizing diagnosis, disease stratification and treatment selection of patients with an inflammatory arthropathy is becoming more widespread92. Ultrasound (US) guided synovial biopsy has proved to be a reliable means to obtain tissue, with many of the early practitioners having once done arthroscopy93. Arthroscopy for synovial biopsy continues at several rheumatologic centers, with the advantages of securing directly visualized tissue, obtaining larger amounts of tissue, and assessing the relationship of synovium to other intraarticular structures. While the skills and costs necessary to perform arthroscopy exceed those for an US procedure, arthroscopic synovial biopsy was shown to be at least as well tolerated as either of the US-guided biopsy procedures; even though arthroscoped patients reported higher levels of pre-procedure pain, patient reports of post-procedure pain and willingness to undergo a repeat biopsy were in the same ranges whether the biopsy was arthroscope or US-guided94.
Technical advances have had a big part in driving progress in arthroscopy. The search for a real office-based “needle scope” came closer to fruition in 2015, when Trice Medical https://tricemedical.com/mi-eye/ and VisionScope https://visionscope-tech.com/ introduced < 2 mm scopes with enhanced imaging processing capability, equivalent to conventional glass lens arthroscopes95. Recently, Arthrex has introduced a similar instrument https://www.arthrex.com/tags/nanoscope. The technology that makes the images from a cell phone camera of sufficient quality to supplant the single-lens reflex camera for most also renders the view from a tiny scope that can be inserted through a 1.4-mm needle puncture a reintroduction to the wonders of arthroscopic inspection, with optics far superior to those of the 1990s era “office arthroscopes.” A cost-benefit analysis assessing the use of the new mini-scope to assess intraarticular pathology in lieu of magnetic resonance imaging found such use would save $115 million–$177 million/yr from a more accurate diagnosis of medial meniscal lesions96. The orthopedic community had begun to recognize the potential of these new smaller arthroscopes97. However, there remains skepticism that, after nearly 50 years of availability with 4 different instruments, needle arthroscopy in the office can duplicate the accuracy of an OR procedure or provide any advantage over MRI other than. the speed of performance98. For the rheumatologist in his/her office, the mini-scope would offer a look at the same range of cartilage and synovial pathologies wondered at by the first rheumatologist to pick up the tool half a century ago. Ike and Kalunian saw this new ‘scope as an avenue for rheumatologists to re-enter the field, expanding on observations already made99. However, neither of our institutions would support the endeavor, citing particular opposition from orthopedic surgeons. When rheumatologic arthroscopy pioneer Nathan Wei died in 2018, Trice lost its most vocal champion in rheumatology and they have had no inquiries from rheumatologists since.
Rheumatologists considering arthroscopy face a different climate now than in the mid-80s when the first real surge of interest began. Diagnostic imaging is so accurate (MRI) for soft tissue and so available at the point of care (ultrasound). Modern therapeutics have obliterated synovectomy as a procedure. None of the soft tissue interventions for OA proved to be successful. And orthopedists facing a shrinking patient base100 hold ever more tightly to their turf (Figure 4).
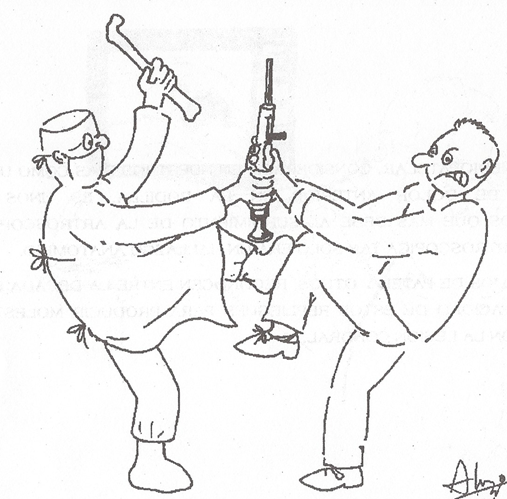
Figure 4: Checa’s cartoon of the orthopedist and rheumatologist wrestling over the arthroscope. From Checa A and Lazo A. Artoscopia. Pasajies y memorias. Havana: CIMEQ press hospital, 1996, with permission.
Arthroscopy has survived as a tool for the rheumatologist almost solely because of its utility in research. There is no duplicate to the view it provides of intraarticular structures, with sampling of those structures superior to what can be done with indirect methods as one sees what is sampled, can appreciate relation of the viewed pathology to other structures, see what is being retrieved, and retrieve as much tissue as desired from a comfortable subject. With the accompanying lavage, therapeutic benefit is possible. Popularity of ultrasound-guided biopsy, which has mostly supplanted arthroscopy, can be understood in terms of ease of performance, lower cost, absence of “turf” issues and wider availability. However, the newest generation of arthroscopes significantly diminishes the cost and “turf” concerns. And the still underappreciated issue of calcinosis in OA cannot be addressed with ultrasound.
Our informal international survey found that pockets of rheumatologists doing arthroscopy still exist, with active centers in Dublin, Leeds, Amsterdam, Barcelona, Lisbon, New Delhi, and Adelaide. There hasn’t been a publication about arthroscopy from a rheumatologist for over two years. Mention of arthroscopy has slid into a line or two in methods sections. While interest in rheumatology is on the rise among U.S. Internal Medicine residents, a taste for the hands-on procedural aspects of the specialty is not, at least by our observations. The ancient rheumatologist-arthroscopists writing this paper lament this loss of interest in something that was once so popular.
We liken the few existing active centers to the monasteries in the Dark Ages, where the light of learning was kept alive eventually to emerge in the Renaissance101. We lend them our strongest encouragement in their endeavors. The light must return. There is so much left to do.
Conflict of interests
The authors have no conflicts to disclose.
References
- Jackson RW. A history of arthroscopy. Arthroscopy. 2010; 26(1): 91-103.
- Kieser CW, Jackson RW. Severin Nordentoft: The first arthroscopist. Arthroscopy. 2001; 17(5): 532-5.
- Burnam MS. Arthroscopy or direct visualization of joints. J Bone Joint Surg. 1931; 13: 669-95.
- Finkelstein H, Mayer l. The arthroscope. A new method of examining joints. J Bone Joint Surg. 1931; 13: 583-8.
- Burnam MS, Finkelstein H, Mayer L. Arthroscopy of the knee joint. J Bone Joint Surg. 1934; 16: 255-68.
- Geist ES. Arthroscopy: Preliminary report. Journal-Lancet. 1926; 46: 306-7.
- Kieser C . [Ernst Vaubel M. D. German pioneer in the field of arthroscopy 1902-1989] Unfallchirurg 2000;103(1):93-7. [in German]
- Sommer R. Die endoskopie des kniegelenkes. [The endoscopy of knees]. Zentralbl Chir. 1937; 64: 1692-7. [In German]
- Hurter E. Revue de chirugoe orthopédique et reparatrice de l’appareil moteur: [Arthroscopy: a new method of knee examinations]. Revue d’Orthopédie. 1955; 4(5-6): 763-6. [In French]
- Imbert R. L’arthroscopie du genou, sa technique [Arthroscopy of the knee; its technic]. Mars Chir. 1956; 8(3): 368-9. [Article in French]
- Watanabe M. Memories of the early days of arthroscopy. Arthroscopy. 1986; 2(4): 209-14.
- Watanabe M. Articular pumping. J Jap Orthop Assoc. 1949; 24: 30-42. [In Japanese]
- Watanabe M. Atlas of Arthroscopy. Tokyo: Igaku Shoin Ltd. 1957.
- Watanabe M, Takeda S, Ikeuchi H. Atlas of Arthroscopy. 2nd edition. Tokyo: Igaku Shoin Ltd, 1969.
- Takeda S. (Relationship between arthroscopic and histological observations for the synovial membrane). J Jap Orthop Assoc. 1960; 34: 373. [in Japanese]
- Gil JR, Katona G. Arthroscopy as a means of diagnosis and research: review of 80 arthroscopies. Proceedings of the Fourth Pan American Congress of Rheumatology. Mexico City. Excerpta Med Intern Congress. 1967; 165: 209-15.
- Torres-Moya R. La artroscopia en Cuba, sus inicios en el Servicio de reumatología del Hospital Docente Clínico Quirúrgico 10 de Octubre [Arthroscopy in Cuba, its beginnings in the Rheumatology Service of the Teaching Clinical Surgical Hospital October 10]. Revista Cubana de Reumatologia. 2013; 13: 1-3. [in Spanish]
- Marques J, Santamaria A, Gomez Martinez G, et al. [Arthroscopy]. Rev Esp Reum Enferm Osteoartic. 1971; 14(1): 47-58. [in Spanish]
- Andersen RB, Rossel I. [Arthroscopy of the knee joint in rheumatic diseases]. Ugeskr Laeger. 1973; 135(2): 71-3. [in Danish]
- Dorfman H, de Seze S. Nouvelles observations sur I'arthroscopie du genou. Resultat d'une experience personelle. [New observations on knee arthroscopy. Result of a personal experience]. Semaine des Hopitaux de Paris. 1972; 48: 3011-9. [In French]
- Marques J, Barcelo y GP. La artroscopia. [The arthroscopy]. Barcelona: Pher Laboratories, 1977. [in Spanish]
- Griego JG. La artroscopia de la rodilla. Experiencia de un reumatólogo. [The arthroscopy of the knee. Experience of a rheumatologist]. ECIMED, Ciudad de la Habana, 1991. [in Spanish]
- Jackson RW. Memories of the early days of arthroscopy: 1965-1975. The formative years. Arthroscopy. 1987; 3(1): 1-3.
- Altman RD: Arthroscopic findings of the knee in patients with pseudogout. Arthritis Rheum. 1978; 19: 286-92.
- Altman RD, Gray R. Diagnostic and therapeutic uses of the arthroscope in rheumatoid arthritis and osteoarthritis. Am J Med. 1983 Oct 31; 75(4B): 50-5.
- Jayson MIV, Dixon AS. Arthroscopy of the knee in rheumatic diseases. Ann Rheum Dis. 1968; 27: 503-11.
- Yates DB, Scott JT. Rheumatoid synovitis and joint disease. Ann Rheum Dis. 1975; 34: 1-6.
- Fletcher MR, Scott JT. Chronic monarticular synovitis: diagnostic and prognostic features. Ann Rheum Dis. 1975; 34: 7-6.
- Yates DB. Arthroscopy, arthrography and synovial biopsy. In Copeman's Textbook of the Rheumatic Diseases, 5th edn. 1978; 1022-1031. Edited by J. T. Scott, Churchill Livingstone: London and New York. Continued into 6th edn (’88) 1597-1611, last edition
- Dandy DJ, Jackson RW. The diagnosis of problems after meniscectomy. J Bone Jt Surg [Br]. 1975; 57: 346-8.
- Jackson DW. Videoarthroscopy: a permanent medical record. Am J Sports Med. 1978; 6(5): 213-6.
- Johnson LL. Arthroscopic Surgery: Principles and Practice. St. Louis, C. V. Mosby, 1986.
- Stulberg SD, Keller CS. The principles and results of arthroscopic surgical treatment of rheumatoid arthritis of the knee. Arthritis Rheum. 1981; 24: S128.
- Kelley WN. A new role for the ARA in guiding our destiny. Arthritis Rheum. 1987; 30(11): 1201-4.
- Lindblad S, Hedfors E. Intraarticular variation in synovitis. Local macroscopic and microscopic signs of inflammatory activity are significantly correlated. Arthritis Rheum. 1985; 28(9): 977-87.
- Frassica FJ, Combs JJ, Sim FH. Synovial proliferative disorders: differential diagnosis. Arthroscopy. 1985; 1(3): 183-9.
- Arnold WJ, Kalunian K. Arthroscopic synovectomy by rheumatologists: time for a new look. Arthritis Rheum. 1989; 32(1): 109-11.
- Morris AH, Jennings JE, Stone RG, et al. Guidelines for privileges in arthroscopic surgery. Arthroscopy. 1993; 9: 125–7.
- Chang RW, Falconer J, Stulberg SD, et al. A randomized, controlled trial of arthroscopic surgery versus closed-needle joint lavage for patients with osteoarthritis of the knee. Arthritis Rheum. 1993; 36(3): 289-96.
- Chang RW, Sharma L. Why a rheumatologist should be interested in arthroscopy. Arthritis Rheum. 1994; 37: 1573–6.
- Mund DJ. Why a practicing rheumatologist might not be interested in arthroscopy: comment on the editorial by Chang and Sharma. Arthritis Rheum. 1994; 37: 1026.
- Combe B, Krause E, Sany J. Treatment of chronic knee synovitis with arthroscopic synovectomy after failure of intraarticular injection of radionuclide. Arthritis Rheum. 1989; 32(1): 10-4.
- Arnold WJ. Office-based arthroscopy. Bull Rheum Dis. 1992; 41(5): 3-6.
- Halbrecht JL, Jackson DW. Office arthroscopy: a diagnostic alternative. Arthroscopy. 1992; 8(3): 320-6.
- Reece R, Emery P. Needle arthroscopy. Br J Rheumatol. 1995; 34(12): 1102-4.
- Ike RW, O'Rourke KS. Detection of intra-articular abnormalities in osteoarthritis of the knee: A pilot study comparing needle arthroscopy with standard arthroscopy. Arthritis Rheum. 1993; 36: 1353-63.
- Denti M, Arosio A, Trevisan C. Comparison of “catheter” and conventional arthroscopy in the diagnosis of knee derangements. Arthroscopy. 1994; 10(6): 614-7.
- Meister K, Harris NL, Indelicato PA, et al. Comparison of an optical catheter office arthroscope with a standard rigid rod-lens arthroscope in the evaluation of the knee. Am J Sports Med. 1996; 24(6): 819-23.
- Ike RW, Fox DA. Arthroscopy in rheumatology training programs associated with NIH-multipurpose arthritis centers: Results from a survey of program directors. Arthritis Rheum. 1993; 35: 1329-31.
- Kalunian KC, Moreland L, Ike RW. Arthroscopy, in Arthritis and Allied Conditions, WJ Koopman, Editor. Thirteenth edition. Williams & Wilkins: Baltimore. 1996; 103-114.
- Ike RW. Minimally invasive procedures, in Rheumatology, JH Klippel, PA Dieppe, Editors. Second edition. 1997, Mosby-Wolfe: London. p.2.13.1-2.13.10.
- Baeten D, Van den Bosch F, Elewaut D, et al. Needle arthroscopy of the knee with synovial biopsy sampling: technical experience in 150 patients. Clin Rheumatol. 1999; 18: 434–41.
- Smith MD, Chandran G, Youssef PP, et al. Day case knee arthroscopy under regional anaesthesia, performed by rheumatologists. Aust NZ J Med. 1996;26:108–9.54.
- Michalska M. Knee arthroscopy in the office. Hosp Pract (Off Ed). 1997; 32: 179–82, 184, 190.
- Wei N, Delauter SK, Erlichman MS. Office-based arthroscopy. Evolution of the procedure: the second 100 cases. J Clin Rheumatol. 1995; 1(4): 219-26.
- Ike RW, Arnold WJ. Correlations between radiographic changes, meniscal chondrocalcinosis and other intra-articular abnormalities in patients with osteoarthritis of the knee undergoing arthroscopy. Arthritis Rheum. 1987; 30(l Suppl): S17 and 30(4 Suppl):S13.
- Hunter DJ, Zhang YQ, Niu JB, et al. The association of meniscal pathologic changes with cartilage loss in symptomatic knee osteoarthritis Arthritis Rheum. 2006; 54(3): 795-801.
- Ike RW, O'Rourke KS. Compartment directed physical exam of the knee can predict articular cartilage abnormalities disclosed by needle arthroscopy. Arthritis Rheum. 1995; 38: 917-25.
- Reece RJ, Canete JD, Parsons WJ, et al. Distinct vascular patterns of early synovitis in psoriatic, reactive, and rheumatoid arthritis. Arthritis Rheum. 1999; 42(7): 1481-4.
- Canete JD, Rodriguez JR, Salvador G, et al. Diagnostic usefulness of synovial vascular morphology in chronic arthritis. A systematic survey of 100 cases. Semin Arthritis Rheum. 2003; 32: 378–87.
- Moll C, Bogas M, Gomez-Puerta JA, et al. Macroscopic features of knee synovitis in early untreated Behçet disease and psoriatic arthritis. Clin Rheumatol. 2009; 28(9): 1053-7.
- Ike R, Baskaran A, Chao J, et al. Prevalence of crystalline disease in knee osteoarthritis: experience from patients undergoing arthroscopic procedures. J Clin Rheumatol. 2019; 25(1): 51-3.
- Wei N, Beard S, Bitner C, et al. Osteoarthritis- A Crystal Induced Disease? Unpublished observations 2017.
- Ayral X, Dougados M, Listrat V, et al. Chondroscopy: a new method for scoring chondropathy. Semin Arthritis Rheum. 1993; 22: 289-97.
- Ayral X, Gueguen A, Ike RW, et al. Inter-observer reliability of the arthroscopic quantification of chondropathy of the knee. Osteoarthritis Cartilage. 1998; 6: 160–6.
- Ayral X, Dougados M. Rheumatological arthroscopy or research arthroscopy in rheumatology? Br J Rheumatol. 1998; 37: 1039–41.
- Brodenick LS, Turner DA, Renfrew DL, et al. Severity of articular cartilage abnormality in patients with osteoarthritis: Evaluation with fast spin-echo MR vs arthroscopy. Am J Roentgen. 1993; 162(1): 99-103.
- Laing TJ, Ike RW, Griffiths CE, et al. A pilot study of the effect of oral 8-methoxypsoralen and intraarticular ultraviolet light on rheumatoid synovitis. J Rheumatol. 1995; 22: 29-33.
- Wei N, Delauter SK, Erlichman MS. The holmium YAG laser in office based arthroscopy of the knee: comparison with standard interventional instruments in patients with arthritis. J Rheumatol. 1997; 24(9): 1806-8.
- Wei N, Delauter SK, Erlichman MS. More on laser arthroscopic synovectomy. J Clin Rheumatol. 1999; 5(3): 179.
- Chang RW, Falconer J, Stulberg SD, et al. A randomized, controlled trial of arthroscopic surgery versus closed-needle joint lavage for patients with osteoarthritis of the knee. Arthritis Rheum. 1993; 36(3): 289-96.
- Moseley JB, O’Malley K, Petersen NJ, et al. A controlled trial of arthroscopic surgery for osteoarthritis of the knee. N Engl J Med. 2002; 347: 81-8.
- Bradley JD, Heilman DK, Katz BP, et al. Tidal irrigation as treatment for knee osteoarthritis: a sham-controlled, randomized, double-blinded evaluation. Arthritis Rheum. 2002; 46: 100-8.
- Kalunian KC, Concoff AL, Skovron ML, et al. Visually-guided irrigation in early knee osteoarthritis: A multi-center randomized prospective study. Osteoarthritis Cartilage. 2000; 8: 411-8.
- Katz JN, Brownlee SA, Jones MH. The role of arthroscopy in the management of knee osteoarthritis. Best Pract & Res Clin Rheumatol. 2014; 28: 143-56.
- Yousswl P, Breedweld F, Bresnihan B. Quantitative microscopic analysis of inflammation in rheumatoid arthritis synovial membrane samples selected at arthroscopy compared with samples taken blindly by needle biopsy. Arthritis Rheum. 1997; 40(9): 45-9.
- Veale DJ. The role of arthroscopy in early arthritis. Clin Exp Rheumatol. 1999; 17: 37–8.
- Szachnowski P, Wei N, Arnold WJ, et al. Complications of office based arthroscopy of the knee. J Rheumatol. 1995; 22: 1722–5.
- Wollaston S, Brion P, Kumar A, et al. Complications of knee arthroscopy performed by rheumatologists. J Rheumatol. 2001; 28(8): 1871-3.
- Kavanaugh A, Rosengren S, Lee SJ, et al. Assessment of rituximab's immunomodulatory synovial effects (ARISE trial). 1: clinical and synovial biomarker results. Ann Rheum Dis. 2008; 67(3): 402-8.
- Kane D, Veale DJ, FitzGerald O, et al. Survey of arthroscopy performed by rheumatologists. Rheumatology. 2002; 41(2): 210-215.
- Gualco G, Prodanov A, Andujar R, et al. Papel de la biopsia sinovial en reumatología. Análisis de 80 casos Dres. [Role of synovial biopsy in rheumatology. Analysis of 80 Dres cases]. Rev Med Uruguay. 2000; 16: 4-17. [in Spanish]
- Kuzmanova SI, Zaprianov ZN, Solakov PT. Correlations between arthroscopic findings and synovial membrane histology in patients with rheumatoid synovitis of the knee joint. Folia Med (Plovdiv). 2003; 45(3): 60-5.
- Reece RJ. Arthroscopy as a Research Tool. Arthritis Res. 2007; 8(2): 27-45.
- Goeb V, Walsh CAE, Reece RJ, et al. Potential role of arthroscopy in the management of inflammatory arthritis. Clin Exp Rheumatol. 2012; 30: 429-35.
- Wechalekar MD, Smith MD. Arthroscopic guided synovial biopsy in rheumatology: current perspectives. Int J Rheum Dis. 2017; 20: 141-4.
- Vordenbaumen S, Sewerin P, Logters T, et al. Inflammation and vascularisation markers of arthroscopically-guided finger joint synovial biospies reflect global disease activity in rheumatoid arthritis. Clin Exp Rheumatol. 2014; 32(1): 117-20.
- Chaturvedi V, Thabah MM, Ravindran V, et al. Medical arthroscopy: A tool for diagnosis and research in rheumatology. Int J Rheum Dis. 2017; 20: 145-53.
- Sharma A, Baethge BA, Acebes JC, et al. Arthroscopic lavage treatment in rheumatoid arthritis of the knee. J Rheumatol. 1996; 23(11): 1872-4.
- Tanaka N, Sakahashi H, Hirose K, et al. Volume of a wash and the other conditions for maximum therapeutic effect of arthroscopic lavage in rheumatoid knees. Clin Rheumatol. 2006; 25(1): 65-9.
- van Oosterhout M, Sont JK, Bajema IM, et al. Comparison of efficacy of arthroscopic lavage plus administration of corticosteroids, arthroscopic lavage plus administration of placebo, and joint aspiration plus administration of corticosteroids in arthritis of the knee: A randomized controlled trial. Arthritis Rheum. 2006; 55(6): 964-70.
- Orr C, Vieira-Sousa E, Boyle DL, et al. Synovial tissue research: a state-of-the-art review. Nat Rev Rheumatol. 2017; 13(8): 463-475. Erratum in: Nat Rev Rheumatol. 2017; 2213(10 ): 630. Nat Rev Rheumatol. 2017;14 (1).
- Romao VC, Polido-Pereira J, Barros R, et al. Efficacy, safety and sample quality of ultrasound-guided synovial needle biopsy in clinical practice and research: a prospective observational study. Arthritis Care Res (Hoboken). 2019 Aug 17. doi: 10.1002/acr.24050. [Epub ahead of print]
- Just SA, Humby F, Lindegaard H, et al. Patient-reported outcomes and safety in patients undergoing synovial biopsy: comparison of ultrasound-guided needle biopsy, ultrasound-guided portal and forceps and arthroscopic-guided synovial biopsy techniques in five centres across Europe. RMD Open. 2018; 4(2): e000799. doi: 10.1136/rmdopen-2018-000799.
- McMillan S, Saini S, Alyea E, et al. Office-Based Needle Arthroscopy: A Standardized Diagnostic Approach to the Knee. Arthroscop Tech. 2017; 6(4): e1119-e1124.
- Voigt JD, Mosier M, Huber B. In-office diagnostic arthroscopy for knee and shoulder intra-articular injuries its potential impact on cost savings in the United States. BMC Health Serv Res. 2014; 14: 203.
- Zhang K, Crum RJ, Samuelsson K, et al. In-Office Needle Arthroscopy: A Systematic Review of Indications and Clinical Utility. Arthroscopy. 2019; 35(9): 2709-21.
- McIntyre LF. Editorial commentary: what you see is what you get – is in-office needle arthroscopy ready for prime time? Arthroscopy. 2019; 35(9): 2722-3.
- Ike RW, Kalunian KC. Arthroscopy in rheumatology: time for a new look? J Rheumatol. 2018; 45(3): 300-1.
- Schroeder MO. The case for – and against – arthroscopic knee surgery. U.S. News and World Report. 6/22/17.
- Greenblatt S. The swerve. How the world became modern. New York: W.W. Norton and Company, 2012.
