Fate of the Coronary Ostial and Distal Aortic Anastomoses after Modified Bentall’s Operation (UKC’s Modification)
Ujjwal K. Chowdhury1*, Niwin George1, Sukhjeet Singh1, Suruchi Hasija2, Lakshmikumari Sankhyan1, Srikant Sharma1, Niraj Nirmal Pandey3, Sanjoy Sengupta1, Mani Kalaivani4
1Departments of Cardiothoracic and Vascular Surgery, All India Institute of Medical Sciences, New Delhi, India.
2Department of Cardiac Anaesthesia, All India Institute of Medical Sciences, New Delhi, India.
3Department of Cardiac Radiology ,All India Institute of Medical Sciences, New Delhi, India.
4Department of Biostatistics, All India Institute of Medical Sciences, New Delhi, India.
Abstract
Graphical Abstract
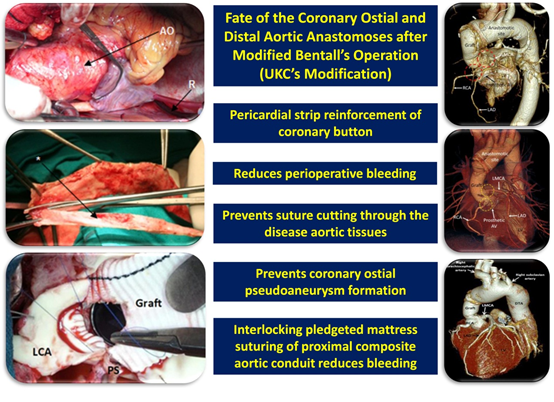
Graphic display (n=140) showing widely patent left and right coronary arterial systems, intact proximal and distal anastomoses without coronary ostial kinking/narrowing/aneurysm or pseudo-aneurysm formation
Objectives: To ascertain the influence of technique of coronary button implantation in patients undergoing Bentall’s procedure with respect to survival, re-exploration for bleeding, anastomotic complications of proximal and distal aortic suture lines, formation of coronary aneurysm and pseudo-aneurysm, kinking and coronary ostial narrowing.
Methods: We reviewed 140 patients (75 males) aged between 22 and 66 years (mean±SD 48.21±1.36 years) receiving a composite aortic conduit from January 1998 to December 2019 for annulo-aortic ectasia (n=98), aortic dissection (n=41), and repaired tetralogy of Fallot (n=1), 24 of whom had Marfan’s syndrome. All patients underwent modified “button technique” by interposing a glutaraldehyde-treated pericardial strip at the graft-coronary anastomoses and proximal aortic conduit suturing using interlocking, interrupted, pledgeted mattress sutures. To detect anastomotic complications of the coronary ostia, proximal and distal aortic anastomoses, survivors underwent echocardiography every 6 months and computed tomographic angiography at 12 months or whenever indicated.
Results: Seven (5%) patients died of cardiac-related cause, 45% had transient hemodynamic instability, 55% had low cardiac output and 87.1% had spontaneous return of sinus rhythm. At a mean follow-up of 161.6±69.9 months, the actuarial survival was 92.6±0.02% (95% CI: 86.9%-96.1) and there were no anastomotic aneurysm or pseudo-aneurysm formation, no kinking of the coronary ostia or ostial narrowing.
Conclusion: The modified open button technique and proximal aortic conduit suturing is associated with reduced perioperative bleeding, no aneurysm or pseudo-aneurysm formation at the coronary and distal aortic anastomoses. We recommend more extensive use of pericardial strip reinforcement of coronary button and new imaging techniques.
Introduction
Long-term results after composite graft aortic root replacement may depend on the implantation technique. The original Bentall’s procedure employed an in situ circumferential suture line around the coronary ostia, then complete aortic wrap around to control bleeding. Tension developing at the suture line of the side-to-side coronary anastomosis and/or blood accumulation within the space around the graft conveyed significant risk of coronary artery dehiscence, pseudo-aneurysm formation and reoperation1, 2. Hematoma in the space may additionally compress the graft or cause prosthetic valve dysfunction1-3.To avoid this complication, Cabrol used a fistula to the right atrial appendage and subsequently adopted interposition Dacron conduits to the coronary ostia with the new risk of graft thrombosis and persistent aorto-right atrial fistula4, 5. For these reasons, a modified open technique has been developed by Kouchoukos and associates in 1986 that basically eliminate the prosthetic wrap and allows reattachment of adequately mobilized coronary buttons to the graft with tension free sutures1.
Despite the introduction of these technical modifications, complications after composite graft replacement of the ascending aorta have not been eliminated. Problems encountered with these techniques include bleeding from the proximal conduit implant site and reattached coronary artery origins, suture line disruption, coronary artery distortion and late development of pseudoaneurysms6-14. To address these concerns, we interposed a glutaraldehyde-treated autologous pericardial strip between the graft and native coronary ostia during coronary ostial implantation and placed interlocking, interrupted, pledgeted mattress sutures to ensure perfect haemostatic suture lines and published our initial experience in 201715-17.
The objective(s) of this retrospective study were to evaluate: i) the stability of the modified Bentall’s procedure over time; ii) the influence of surgical technique adopted for reattachment of the coronary ostia in the fate of the coronary anastomoses; iii) the occurrence of coronary ostial pseudo-aneurysms, aneurysm of the anastomotic site to the composite graft and aneurysms of the left main trunk and right coronary artery, iv) the occurrence of kinking and narrowing of the coronary ostia during follow-up, and v) the influence of technique of coronary artery implantation on the short and long-term results in composite aortic root replacement.
Patients and Methods
This study conforms to the principles outlined in the declaration of Helsinki. Between January 1998 and December 2019, 140 consecutive patients (80 males), aged 22-66 years (mean 48.21±13.6 years) underwent modified Bentall’s operation using the surgical techniques described by a single surgeon (corresponding author) after obtaining informed consent and institutional ethics committee approval. Patients’ demographic details are shown in table 1.
Table 1: Demographic, operative, and postoperative details of the patients in the study (n=140)
|
Variables |
Number (%) |
|
Mean age at operation (range) |
48.21±13.6 years (22-66 years) |
|
Sex- Males |
75 (53.5%) |
|
Annulo-aortic ectasia |
98 (70%) |
|
Type A aortic dissection |
41 (29.3%) |
|
Repaired tetralogy of Fallot with aortic valvular reconstruction |
1 (0.7%) |
|
Moderate-to-severe aortic regurgitation |
134 (95.7%) |
|
Moderate mitral regurgitation |
6 (4.3%) |
|
Left ventricular ejection fraction <0.40 |
103 (73.6%) |
|
Marfan’s syndrome |
24 (17.1%) |
|
Renal dysfunction |
72 (51.4%) |
|
Concomitant coronary artery bypass grafting |
8 (5.7%) |
|
Operation timing
|
37 (26.4%) 103 (73.6%) |
|
Mean aortic cross-clamp time (range) |
111.8 ± 28.2 (89-186 minutes) |
|
Mean cardiopulmonary bypass time (range) |
154 ± 28.8 (136-218 minutes) |
|
In-hospital mortality |
7 (5%) |
|
Late death |
3 (2.1%) |
|
Mean hospital stay (range) |
19±7 days (6-52 days, median 9 days) |
|
Low cardiac output
|
77 (55%) 63 (45%) |
|
Average postoperative drainage (12 hours) |
242±60 ml |
Surgical Techniques
Intraoperative trans-esophageal echocardiography (TEE) was performed with a Hewlett-Packard Sonos 1500 or 5500 ultrasound system on all patients both before and after surgery. Standard anaesthetic and operative techniques were used throughout the study period. The operations were performed under moderately hypothermic cardiopulmonary bypass (CPB) through arterial cannulation (axillary artery n=28; femoral artery n=112) and bicaval venous cannulation through the femoral vein and superior vena cava.
A retrograde coronary sinus cannula with self-inflatable balloon (RCO 14, Edwards Lifesciences, Irvine, CA, USA) was used in all patients. Trans-atrial blind cannulation of the coronary sinus was performed in 92 patients. In cases of difficult cannulation (n=48), the coronary sinus was cannulated through a short right atriotomy under direct vision. The proper placement was confirmed by observing distension of the posterior inter-ventricular vein, maintenance of coronary sinus pressure, palpation of the coronary sinus cannula posteriorly at the base of the heart and trans-esophageal and TEE16. No anomalies of the coronary sinus or coronary sinus injuries were confronted.
Ultra-filtration was used in all patients during and after CPB to reduce the total body water, potassium overload and to remove the inflammatory mediators from the circulation, maintaining haematocrit >25% on CPB.
All patients were subjected to the same “integrated myocardial preservation management” strategy as was originally proposed by Gerald D, Buckberg18. Intermittent direct ostial, high potassium, cold blood cardioplegia (St Thomas II solution 4:1); containing 27 meq/L potassium at a dose of 150 ml/min/m2 along with topical cooling was used in all patients. After achieving cardiac arrest, the cardioplegia line was switched to the fluid-filled retroplegia cannula, and low potassium cold blood cardioplegia (containing 13 meq/L) was commenced via the auto-inflating coronary sinus cannula at a flow rate of 100-150 ml/min. Coronary sinus pressure was maintained less than 40 mmHg. Antegrade direct ostial coronary cardioplegic infusions (500 ml through both the coronary ostia) were repeated every 20 minutes. Cardioplegic infusions by both routes were never given simultaneously.
A Dacron composite graft with a mechanical heart valve [(St. Jude Medical Inc.; Minn); (Conduit 25 mm, 55 patients; 27mm, 52; 29mm, 33) was used in all patients. While creating the coronary buttons, the surrounding diseased aortic tissue was excised leaving behind a cuff of 10-12mm.
All patients in the study underwent “modified button technique” for reconstruction of the coronary arteries and proximal conduit suturing to obtain perfect hemostasis as developed by the corresponding author (Figures 1A-1I)15,16. The composite graft was sutured to the annulus using everted, interrupted 2-0 sutures over polytetrafluoro-ethylene pledgets. Each interrupted aortic supra-annular sutures were placed in such a way that successive sutures were made to pass through the previous pledget; thus ensuring perfect aortic annular hemostasis (Figures 1A, 1B). Using graft diathermy, two side holes were created on the composite graft measuring around 10mm in diameter in the proposed area of coronary ostial implantation. The left and right coronary buttons were anastomosed in an end-to- side fashion with continuous 5-0 polypropylene suture to openings on the composite graft. While suturing, a long strip of glutaraldehyde treated pericardium, about 1 cm wide was interposed circumferentially around each graft-coronary button anastomosis (Figures 1C-1H). During construction of the inferior portion of the ostial anastomoses, the continuous retrograde perfusion was transiently lowered to avoid flooding and performing perfect haemostatic suturing under vision.
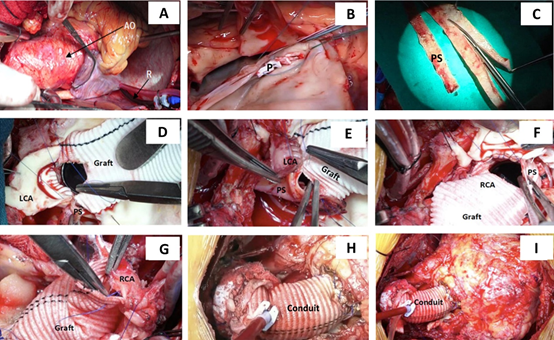
Figures 1A-1I: Surgical photograph of the techniques used for coronary button implantation and proximal aortic conduit suturing in the patient undergoing modified Bentall’s procedure for annulo-aortic ectasia. Figures 1A-1B - note placement of the coronary sinus cannula for continuous retrograde cardioplegia and the placement of interrupted pledgeted mattress sutures for proximal aortic conduit suture line. Each interrupted suture is passed through the previous pledget interlocking with each other, ensuring perfect hemostasis. Figure 1C-1H - step-by-step demonstration of graft-coronary anastomosis. Note the circumferential placement of the pericardial strip between the graft and the native coronary ostia. Figure 1I - the completed placement of the composite aortic valved conduit [AO- Aorta; LA vent- Left atrial vent; LCA- Left coronary artery; PA vent- Pulmonary artery vent; P- Pledget; PS- Pericardial strip; R- Retrograde cardioplegia cannula; RCA- Right coronary artery].
The right coronary button was anastomosed at a relatively high position of the composite graft taking into consideration the collapsed state of the right ventricle during cardioplegic arrest. In cases of large aortic root aneurysm secondary to annulo-aortic ectasia, precautions were taken for implantation of the right coronary artery at an appropriate height to avoid post implant waisting of the right coronary artery. The technical details of the composite aortic graft replacement have been enumerated in detail in our previous publication15. We used topical thrombin (Tisseel, Baxter AG, Vienna) as an additional topical haemostatic agent on all patients.
The left ventricle and main pulmonary artery were vented throughout the cross-clamp period and while performing the distal graft-aortic anastomosis, myocardial perfusion was restored with warm blood administered retrogradely through the coronary sinus catheter. Retrograde perfusion was assured by noting the engorged oxygenated cardiac veins as well as the return of dark blood through the vent in the aortic root.
Following removal of aortic cross-clamp, majority of the patients (87.14%) returned to normal sinus rhythm. Only 18 (12.8%) patients required antegrade administration of injection adenosine and 200-250ml warm hotshot blood cardioplegia for spontaneous defibrillation. Direct current cardioversion was not used on any patient. We used atrioventricular sequential pacing for 24-48 hours in patients with low heart rate.
Statistical Analysis
Statistical analysis was performed using Intercooled STATA 14.0 Software (College Station, Texas, USA). Interval related data were expressed as mean ± standard deviation (SD) or median (minimum-maximum) and categorical variables were expressed as percentages. The Kaplan-Meier curve was drawn to show the probability of survival over a period of follow-up time. The results were expressed as probability of survival (95% confidence interval) at various time intervals. The p value of <0.05 was considered as statistically significant.
Results
Operative mortality
There were seven (5%) early deaths due to LOS with multi-organ failure and superimposed sepsis between 10th and 18th postoperative days. These patients had acute type A aortic dissection and were in congestive cardiac failure (CCF) preoperatively (Table 1).
Morbidity
Hospital morbidity included re-exploration for excessive bleeding from non-anastomotic sites within 12 hours in 8 patients with acute type A aortic dissection. The average 12-hour postoperative drainage was 242±60 ml. Thirteen (9.3%) patients required tracheostomy and long-term ventilator support. Three patients required hospital readmission on 18th, 21st and 22nd days postoperatively because of deranged prothrombin time requiring pericardiocentesis (Table 1).
Long-term outcomes
There were 3 (2.1%) late deaths 15, 60 and 69 months after surgery due to intractable ventricular arrhythmias, anticoagulant-related intracranial bleed and chronic renal failure respectively. Follow-up was 100% complete (1-264 months) and yielded 1750.6 patient-years of data. At a mean follow-up of 161.6 (SD± 69.9) months, the actuarial survival was 92.6±0.02% (95% CI: 86.9-96.1; Figure 2). All survivors were in New York Heart Association I or II at their last follow-up.
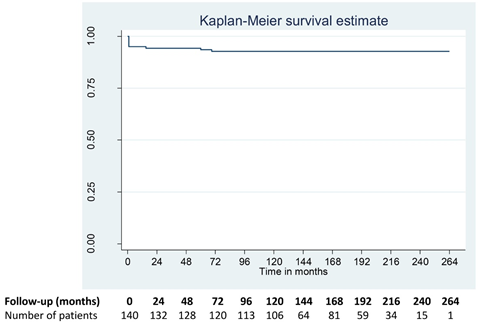
Figures 2: Actuarial survival curve of patients (n=140) undergoing Bentall’s procedure using modified “button technique” and proximal aortic conduit suturing
Postoperative pharmacological and mechanical circulatory support
Patients requiring dopamine and/or dobutamine hydrochloride, 5 µg/kg/min for less than 24 hours postoperatively were considered as having transient post-bypass haemodynamic instability (n=63; 45%). In contrast, patients who required inotropic agents, vasopressors and/or intra-aortic balloon counterpulsation (IABC) for more than 24 hours postoperatively were categorized as having a true LOS (n=77; 55%).
Patients considered to have LOS (n=77) required dopamine (4-10µg.kg-1.min-1), epinephrine (0.01-0.1 µg.kg-1.min-1) and milrinone (50 µg/kg IV bolus followed by 0.375-0.75 µg.kg-1.min-1) either isolated or in combination. Five patients required intra-aortic balloon counterpulsation (IABC) as an additional support. Three patients who required IABC survived. The incidence of LOS remained fairly constant over the course of the study (Definition- Electronics).
Postoperative Hyperkalemia and volume overload
Mean cross-clamp time was 110.8±28.2 min (range, 89-186 minutes) and mean CPB time was 154±28.8 min (range, 136-218 min). The mean flow of cardioplegia in the coronary sinus was 100ml/min (range, 80 to 190 ml/min). Cardioplegic flow rates varied and the total administered volume of retrograde cardioplegia ranged from 3000 ml to 4500 ml. The total amount of cardioplegic solution delivered ranged from 5600 ml to 6500 ml. The total amount of potassium delivered ranged from 88 to 194 meq with a mean of 99 meq. Hyperkalemia was not a clinically significant problem in any patient.
Peri-operative myocardial infarction and arrhythmias
No patient sustained perioperative myocardial infarction. Rhythm abnormalities included supra-ventricular arrhythmias (11.4%), ventricular arrhythmia (7.8%), premature ventricular contraction (n=9), ventricular tachycardia (n=2), bundle branch block (10%) and junctional rhythm (7.1%).
Cohort of survivors
All survivors (n=130) were examined and studied between June 2019 and December 2019, which was the closing interval of the study. Postoperative evaluation consisted of three monthly clinical examination, electrocardiogram, chest radiograph, cine fluoroscopy, echocardiography and computed-tomographic angiography. The functional class at follow-up was noted. All patients received warfarin and aspirin (100 mg/day) for anticoagulation to maintain INR between 2.5 to 3.5. Digoxin and diuretics were weaned at varying time intervals. Amiodarone was used for intractable atrial fibrillation.
The LVEF increased from 49%±9.2% (range, 15% to 62%) to 57%±6% (range, 15% to 70%). Forty-eight (34.3%) patients had LVEF between 15% and 25%.
Computed tomographic (CT) angiography was performed on a third generation, 384 (2 x 192) slice, dual source CT scanner (SOMATOM FORCE, Siemens, Germany). Multi-planar reformatted images and volume rendered images were reconstructed and analysed (Techniques- Electronics).
Aortic sequelae
Persistence of the false lumen in the distal aortic arch or descending aorta without evident dilatation was detected on CT angiography on 32 patients. Three patients required thoraco-abdominal aortic graft replacement with re-implantation of celiac, superior mesenteric and right renal arteries 3 years following Bentall’s procedure. Extension of distal dissection and signs of progressive dilatation of the false lumen was observed in two patients and are currently being evaluated as potential candidates for endovascular stenting. No survivors had structural deteriorations, pseudo-aneurysm formations or thromboembolic complications.
Anastomotic complications
At a mean follow-up of 161.6±65.9 months, none of the survivors demonstrated any anastomotic complications involving the coronary ostia or the distal aortic arch. All patients demonstrated patent coronary ostia and there were no coronary aneurysm, kinking or pseudo-aneurysm formation of the newly implanted coronary arteries (Figure 3).
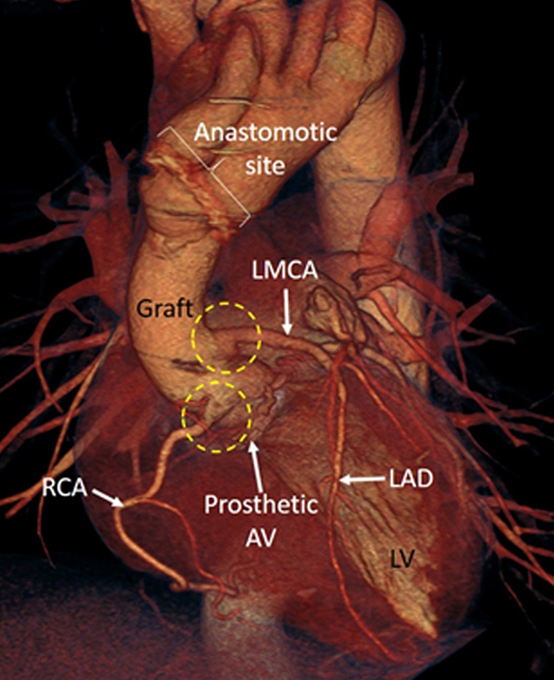
Figures 3: Postoperative computerized volume rendered image in the LAO projection with cranial tilt in this study revealed widely patent left and right coronary arterial systems, intact proximal and distal anastomoses. There are no coronary ostial kinking / narrowing / aneurysm or pseudo-aneurysm formation of the coronary arterial system [Prosthetic AV- Prosthetic aortic valve; DTA- Descending thoracic aorta; LAD- Left anterior descending coronary artery; LCX- Left circumflex artery; LMCA- Left main coronary artery; LV- Left ventricle]
Discussion
Replacement of the ascending aorta and aortic valve with a composite conduit is a well-established method to treat patients with aortic valve disease and aneurysm of the aortic root as well as acute aortic dissection1-7,19.
Wrapping the aortic graft after a Bentall’s procedure has a higher mortality rate and a greater risk of pseudo-aneurysm development, as reported by Kouchoukos and other investigators2,20. Although the button button technique remains the standard technique, the Cabrol repairs are prone to intraoperative occlusion of the right coronary artery and on late follow-up, thrombosis of the right coronary artery is a well-known postsurgical hazard.
Cabrol procedure allows performance of a tension free anastomosis and is still considered a “first line” approach in specific clinical conditions e.g. in cases of reoperation, coronary ostial classification, difficult mobilization of the coronary arteries, extreme aortic dilatation and aortic dissection involving the coronary ostia1. Other indications of Cabrol technique are in coronary or aortic valve intervention in presence of chronic degeneration or familial hypercholesterolemia which leads to porcelain aorta21,22.
Despite the introduction of the open technique by Kouchoukos and associates, postoperative bleeding can still be a major issue being difficult to control once the insertion of the conduit is completed1,23. In order to minimize postoperative bleeding, investigators have introduced several technical modifications into the “open button technique”. These include: i) harvesting of the coronary ostia with a large portion of aortic wall, thus allowing coronary buttons to be sutured in a two-layer fashion (the endo-button buttress technique of Northrup)24; ii) use of an additional horizontal suture line, anchoring the remnant of proximal aorta (8-9 mm above the coronary ostia anastomoses) to the conduit wall (a collar technique of Nezic and associates)25; iii) use of both aortic and supra-annular aortic wall tandem suture lines as described by Copeland23; iv) placement of a long piece of poly-tetra-fluoro-ethylene felt all around the aortic annulus as well as circumferentially around each coronary button anastomoses along with fibrin glue (Sokullu et al)26; and v) placement of an additional continuous suture at the proximal graft anastomosis, i.e. an initial interrupted mattress suture followed by reinforcement suture joining the incised edge of the aortic wall and the prosthetic sewing ring27,28.
Teflon reinforcement of the aortic root has been used by several investigators in operations for acute type A aortic dissection. However, the Teflon felt was not used as an outer supportive layer; instead, the felt was placed between the dissected layers of the aorta to form a neo-intima.
In order to improve haemostasis, Luciani and co-workers have used pericardial strip reinforcement to buttress the inflow (annular) and outflow (ascending aortic) suture lines during Ross operation. These investigators considered the absence of pericardial strip buttressing as an important predictor of auto-graft dilatation28.
Increased CPB time have been associated with an increased risk of mortality following Bentall’s procedure1-6. In a retrospective study of 348 patients, Svensson and associates observed that the risk of early mortality in patients with CPB time exceeding 198 min is five times more than patients undergoing bypass for 65 min or less2.
In the current study, the mean duration of CPB was 154.8±28.8 minutes (range, 136-218 minutes) and mean cross-clamp time was 110.8±28.2 minutes (range, 89-186 minutes). The average volume of postoperative drainage was 242±60 ml in the first 12 hours. In a modification of the Bentall’s procedure, Della Corte and associates used a combination of imbricated proximal suture-line stitches followed by fibrin-sealant spraying to improve hemostasis. Despite efforts to reinforce the sutures and seal the sites of oozing, these investigators reported that the total bleeding volume in the first and second postoperative day amounted to >1000 mL, which is significantly higher than the amount of blood loss observed here30.
Analysis of the hospital mortality data between the classical Bentall, Cabrol and button re-implantation methods in various published series demonstrates almost similar early postoperative outcome1-6. Although Svensson and colleagues suggest that the button technique has better long-term survival with a lower reoperation rate, Kouchoukos, a proponent of button technique reported a series of re-operative root operations where 19% of patients had a Cabrol coronary conduit1-6.
We have analysed the published literature in patients with aortic dissection undergoing Bentall’s procedure by graft inclusion or open button technique. Our results of re-exploration (5.7%) are in accordance with the published data, which documents a re-exploration rate for haemorrhage between 7.2%-21% and 4.6%-19% with the inclusion and open button techniques, respectively1-6,31,32. We believe that the relatively low rate of reoperation for bleeding in our study was achieved due to interposition of the glutaraldehyde-treated pericardial strip between the graft and the native coronary ostia as a buttressing material, thus avoiding sutures cutting through the diseased and friable aortic tissue. We avoided the use of Teflon as a reinforcement material around the proximal aortic suture line and around the coronary ostia, thus avoiding peri-anastomotic extensive fibrous tissue reaction.
The incidence of a coronary ostial pseudo-aneurysm when using a button technique varies from 3.1% to 9%1,7. Recently, Milano, Meijboom and associates have demonstrated that coronary ostial aneurysms are a common finding in patients with Marfan’s syndrome after composite graft replacement even after utilization of the button technique. Coronary ostial aneurysms may also develop after composite graft replacement of the ascending aorta33,34.
At a mean follow-up of 49±19 months, Milano and associates reported anastomotic complications in 6% (4 of 71) patients. Two patients had pseudo-aneurysm at the distal aortic suture line, one patient had pseudo-aneurysm at the right coronary ostium and one Marfan patient had an aneurysm of the left coronary ostium35.
In 2002, Meijboom and associates have demonstrated that, despite using the button technique, coronary ostial aneurysms were detected in 17 of 40 patients undergoing modified Bentall’s procedure utilising “Button technique”. Among Marfan patients aged less than 35 years undergoing Bentall’s procedure, coronary ostial aneurysms were seen in 56% of patients8. Other investigators documented a pseudo-aneurysm formation in 3 of 15 patients who underwent control angiography 6 to 58 months after operation. Niederhauser and associates reported a specific coronary ostia pseudo-aneurysm formation in 3% of their patients.
Gott reported 270 aortic root replacement, in both Marfan’s and non-Marfan’s syndrome, utilizing the classical Bentall’s technique. They reported 6 (2.5%) coronary false aneurysms. Although anastomotic pseudo-aneurysms have been reported in patients with aortic dissection and in Marfan patients by other investigators, we have not noticed the same in our patient population19.
Okamoto and associates classified coronary ostial aneurysms into 3 categories: pseudo-aneurysm, aneurysm of the anastomosis site to the composite graft and aneurysm of the left main trunk itself9. Anastomotic pseudo-aneurysms were observed only in patients with acute dissection or Marfan’s syndrome9,35. The aetiology of true aneurysm of the left main coronary artery is cystic medio-necrosis.
According to Kazui and associates in order to reduce the risk of aneurysm formation at the coronary button anastomosis, the size of the side hole on the composite graft must be reduced to fit the diameter of the coronary artery ostium and avoid the native aortic wall from developing aneurysmal changes in the late postoperative period34. Generally, the side hole measures around 10 mm in diameter. Leaving too much native aortic tissue in a too large opening in the graft seems to be the main cause responsible for coronary ostial aneurysm. The development of an aneurysm in the left main stem itself in the presence of a normal ostium is uncommon.
The anastomotic pseudo-aneurysm and true aneurysm involving the coronary ostia or graft aortic junction could be potentially lethal when it grows to a large diameter. Secondly, irrespective of the size, the aneurysms are known to rupture in equal frequency12.
The utility of new imaging techniques in evaluating patients undergoing Bentall’s procedure has been reinforced by several investigators including ourselves. In 1999, Fattori and associates have demonstrated that MRI is a useful investigative modality to study the morphology of the composite grafts and re-implanted coronary arteries and have resulted in increased detection of anastomotic complications12-17. In this study group, three patients of aortic dissection were re-operated and two patients are awaiting endovascular stenting. None of them had any anastomotic or pseudo-aneurysm.
Technical problems still cause mortality from perioperative myocardial infarction, right ventricular dysfunction and bleeding. With impervious vascular grafts, wraparound of the native aorta is a redundant and potentially troublesome manoeuvre. We achieve better access to potential bleeding sites by complete aortic transection and open button technique.
In a study on 140 aortic root replacements, Westaby and colleagues concluded that right ventricular dysfunction and perioperative myocardial infarction usually occur through technical difficulties with right coronary implantation. They recommended temporarily filling the conduit after performing the distal conduit anastomosis prior to right coronary implantation. Tension or kinking of the anastomosis is avoided with an appropriate extension technique using a wedge of pericardium to augment the superior circumference20. Some investigators prefer using a Dacron interposition graft. Replacement of the aortic root with a composite graft can be performed with an overall early mortality ranging between 5% and 18%1-6. In the present study, our overall early mortality is 9.3%, which is in accordance with the above cited results.
In this study, among non-dissection patients, the early mortality is 5.4%, whereas in acute aortic dissection, the early mortality is 24.2% (p=0.001) comparable to other published series1-6. In an attempt to reduce postoperative adverse cardiac events and improved biventricular performance and survival, we embarked on a program of “integrated myocardial protection” in patients undergoing Bentall’s procedure and published our preliminary observation on 130 patients in 201916. We demonstrated that a combination of “intermittent antegrade and continuous retrograde cold blood cardioplegia” during Bentall’s operation coordinates the myocardial protective strategies with uninterrupted surgical procedure, provides unimpaired vision, avoid unnecessary ischemia, improves ventricular performance and permits aortic unclamping and discontinuation of bypass after surgery16.
At a mean follow-up of 119.8±67.13 months, the actuarial survival was 93.6%±0.02% and there were no extrinsic coronary artery compression, coronary aneurysm of pseudo-aneurysm formation, no coronary ostial narrowing, no anastomotic complications of proximal or distal aortic suture lines and no prosthetic valve endocarditis. Only 12 (8.6%) patients needed 3 days or more to stay in the ICU due to hemodynamic or respiratory problems. The mean duration of hospital stay was 19±7 days (range, 6-52 days).
At a mean follow-up of 119.8±67.13 months, the actuarial survival was 93.6%±0.02% and there were no extrinsic coronary artery compression, coronary aneurysm of pseudo-aneurysm formation, no coronary ostial narrowing, no anastomotic complications of proximal or distal aortic suture lines and no prosthetic valve endocarditis. Only 12 (8.6%) patients needed 3 days or more to stay in the ICU due to hemodynamic or respiratory problems. The mean duration of hospital stay was 19±7 days (range, 6-52 days).
We believe that some important trends have occurred in our experience with the techniques described here-in. First, achievement of perfect haemostasis of the proximal aortic annular suture line by placing interrupted, interlocking pledgeted mattress sutures. Second, although the Bentall’s repair is prone to development of false aneurysms at the coronary anastomoses, we did not observe any aneurysm or pseudo-aneurysm in our study population. We believe that interposition of a glutaraldehyde treated autologous pericardial strip as a buttressing material while re-implanting the coronary arteries to the neo-aortic location may have reduced the physical stress against the native wall of the fragile and diseased aortic tissue surrounding the coronary sinuses. This may have reduced/eliminated the chances of suture cutting through causing intraoperative bleeding and/or later pseudo-aneurysm formation.
We also advise that all patients be evaluated every 6 months to yearly by computed tomography, magnetic resonance imaging or echocardiography to detect the development of false aneurysms or progression of more distal disease, particularly in patients with Marfan’s syndrome or acute dissection.
Study Limitations
The study has several limitations. In as much as this is a single-surgeon, single center study, the results may not necessarily be applicable to all centres. Secondly, the aneurysmal changes may spread overtime particularly in patients with Marfan’s syndrome. Thirdly, the study population is heterogenous. The cases of aortic dissection and Marfan’s syndrome are distinctly different aortic tissues but they have been evaluated together in this investigation. Fourthly, we have not compared the glutaraldehyde treated pericardial strip interposition technique with other materials e.g. Teflon strip. The influence of our technical modifications in preventing anastomotic complications need to be validated on longer follow-up and possibly in prospective randomized studies using other materials for reinforcement of coronary buttons. Future research should focus in defining type and intensity of follow-ups and provide anatomic and flow characteristics to enable comparisons between different modifications of the Bentall’s technique.
Conclusions
Our results confirm that the modified Bentall’s procedure is a valid option for patients who require simultaneous replacement of the aortic valve and ascending aorta with no incidence of coronary ostial pseudo-aneurysm with our modification of coronary button implantation at long-term follow-up. Secondly, the openings in the Dacron graft for implantation of the coronary artery in Marfan’s patients should be kept relatively small to reduce the amount of diseased aortic sinus tissue at the implant sites. This communication is not meant in any way to convince those surgeons satisfied with their own surgical techniques of performing modified Bentall’s operation. Rather it hopes to point out that the techniques described herein provided satisfactory results including reduced cardiopulmonary bypass and aortic cross-clamp time and reduced perioperative bleeding, pseudo-aneurysms, coronary events and prosthesis related complications were not observed.
References
- Kouchoukos NT, Marschall WG, Wedige-Stecker TA. Eleven year experience with composite graft replacement of the ascending aorta and aortic valve. J Thorac Cardiovasc Surg. 1986; 92: 691-705.
- Svenson L, Crawford ES, Hess KR, et al. Composite valve graft replacement of the proximal aorta: comparison of techniques in 348 patients. Ann Thorac Surg. 1992; 54: 427-39.
- Coselli JS, Crawford ES. Composite valve-graft replacement of aortic root using separate Dacron tube for coronary artery reattachment. Ann Thorac Surg. 1989; 47(4): 558-65.
- Cabrol C, Pavie A, Gandjabakhch I, et al. Complete replacement of the ascending aorta with re-implantation of the coronary arteries: new surgical approach. J Thorac Cardiovasc Surg. 1981; 81: 309-315.
- Mestres CA, Betriu A, Pomar JL. Occlusion of the coronary perfusion graft: an exceptional complication after reconstruction of the ascending aorta with the Cabrol technique. J Thorac Cardiovasc Surg. 1989; 98: 302-303.
- Kouchoukos NT. Inclusion (aneurysm wrap) technique for composite graft replacement of the ascending aorta and aortic valve [Letter]. J Thorac Cardiovasc Surg. 1988; 96: 967.
- Yakut C. A new modified Bentall procedure: the flanged technique. Ann Thorac Surg. 2001; 71(6): 2050-2.
- Meijboom LJ, Nollen GJ, Merchant N, et al. Frequency of coronary ostial aneurysms after aortic root surgery in patients with the Marfan syndrome. Am J Cardiol. 2002; 89: 1135-1138.
- Okamoto K, Casselman FP, De Geest R, et al. Giant left coronary ostial aneurysm after modified Bentall procedure in a Marfan patient. Interact Cardiovasc Thorac Surg. 2008; 7: 1164-1166.
- Milano VD, Pratali S, Mecozzi G, et al. Fate of coronary ostial anastomoses after the modified Bentall procedure. Ann Thorac Surg. 2003; 75: 1797-1802.
- Grapow MT, Matt P, Zerkowski HR, et al. Pseudo-aneurysm in a Marfan patient 16 years after Bentall operation. Asian Cardiovasc Thorac Ann. 2004; 12: 182-183.
- Fattori R, Descovich B, Bertaccini P, et al. Composite graft replacement of the ascending aorta: leakage detection with gadolinium-enhanced MR imaging. Radiology. 1999; 212: 573-577.
- Mesana TG, Caus T, Gaubert JY, et al. Late complications after prosthetic replacement of the ascending aorta: what did we learn from routine magnetic resonance imaging follow-up? Eur J Cardiothorac Surg. 2000; 313-20.
- Dossche KM, Schepens MAAM, Morshuis WJ, et al. A 23 year experience with composite valve graft replacement of the aortic root. Ann Thorac Surg. 1999; 67: 1070-1077.
- Chowdhury UK, Rao K, Narang R, et al. An alternative technique of coronary button implantation in patients undergoing modified Bentall’s procedure. Journal of Cardiac Critical Care. TSS 2017; 1(1): 51-54.
- Chowdhury UK, George N, Sankhyan LK, et al Intermittent antegrade and continuous retrograde coronary sinus cold blood cardioplegia to prevent and reverse ischemic and reperfusion damage in patients undergoing Bentall’s procedure: A clinical report on 130 patients. International Journal of Clinical Case Studies and Reports. 2019; 1(2): 23-32.
- Chowdhury UK, George N, Gudala V, et al. Bentall’s procedure for annulo-aortic ectasia and severe aortic regurgitation in a patient with repaired tetralogy of Fallot and aortic valvular reconstruction. World Journal of Pediatric and Congenital Heart Surgery. 2019; 10(5): 648-650.
- Buckberg GD. Update on current techniques of myocardial protection. Ann Thorac Surg. 1995; 60: 805-814.
- Gott VL, Greene PS, Alejo DE, et al. Replacement of the aortic root in patients with Marfan’s syndrome. N Engl J Med. 1999; 340: 1307-1313.
- Westaby S, Katsumata T, Vaccari G. Aortic root replacement with coronary button re-implantation: low risk and predictable outcome. Eur J Cardiothorac Surg. 2000; 17(3): 259-65.
- Grenon SM, Lachapelle K, Marcil M, et al. Surgical strategies for severe calcification of the aorta (porcelain aorta) in two patients with homozygous familial hypercholesterolemia. Can J Cardiol. 2007; 23(14): 1159-61.
- Kourliouros A, Soni M, Rasoli S, et al. Evolution and current applications of the Cabrol procedure and its modifications. Ann Thorac Surg. 2011; 91(5): 1636-41.
- Copeland J III, Rosado L, Snyder S. New technique for improving hemostasis in aortic root replacement with composite graft. Ann Thorac Surg. 1993; 55: 1027-1029.
- Northrup W, Kshettry V. Implantation technique of aortic homograft root: emphasis on matching the host root to the graft. Ann Thorac Surg. 1998; 66: 280-284.
- Nezic D, Cirkovic M, Knezevic A, et al. Modified Bentall procedure- ‘a collar technique’ to control bleeding from coronary ostia anastomoses. Interact Cardiovasc Thorac Surg. 2008; 7(4): 709-711.
- Sokullu O, Sanioglu S, Orhan G, et al. New use of teflon to reduce bleeding in modified Bentall’s operation. Tex Heart Inst J. 2008; 35(2): 147-51.
- CopelandJG III, Rosado LJ, Snyder SL. New technique for improving hemostasis in aortic root replacement with composite graft. Ann Thorac Surg. 1993; 55: (4): 1027-1029.
- Pratali S, Milano A, Codecasa R, et al. Improving hemostasis during replacement of the ascending aorta and aortic valve with a composite graft. Tex Heart Inst J. 2000; 27(3): 246-9.
- Luciani GB, Casali G, Tomezzoli A, et al. Recurrence of aortic insufficiency after aortic root remodeling with valve preservation. Ann Thorac Surg. 1999; 67: 1849–52.
- Della Corte A, Baldascino F, La Marca F, et al. Haemostatic modifications of the Bentall procedure: imbricated proximal suture and fibrin sealant reduce postoperative morbidity and mortality rates. Tex Heart Inst J. 2012; 39(2): 206-10.
- Panos A, Amahzoune B, Robin J, et al. Influence of technique of coronary artery implantation on long-term results in composite aortic root replacement. Ann Thorac Surg. 2001; 72: 1497-1501.
- Niederhauser U, Rudiger H, Vogt P, et al. Composite graft replacement of the aortic root in acute dissection. Eur J Cardiothorac Surg. 1998; 13: 144-50.
- Taniguchi K, Nakano S, Matsuda H, et al. Long-term survival and complications after composite graft replacement for ascending aortic aneurysm associated with aortic regurgitation. Circulation 1991; 84(Suppl 5): III31-III39.
- Kazui T, Yamashita K, Terada H, et al. Late reoperation for proximal aortic and arch complications after previous composite graft replacement in Marfan patients. Ann Thorac Surg. 2003; 76: 1203-1207; discussion 1027-1028.
- Milano AD, Pratali S, Mecozzi G, et al. Fate of coronary ostial anastomoses after the modified Bentall procedure. Ann Thorac Surg. 2003; 75: 1797–1801.
Definition (Electronics)
Low output syndrome (LOS)
Low output syndrome following Bentall’s procedure was diagnosed if the patient required inotropic support (dopamine at 4-10 µg.kg-1.min-1), dobutamine at 5-10 µg.kg-1.min-1, epinephrine at 0.01-0.1 µg.kg-1.min-1 either isolated or in combination in the operating room or in the intensive care unit, to maintain stable hemodynamics (systolic blood pressure at greater than 90 mmHg and cardiac output greater than 2.2 L.min-1.m-2) in the absence of residual structural lesions and mechanical external compression after correction of all electrolytes or blood gas abnormalities and after adjustment of the preload to its optimal value. Low-output syndrome was also diagnosed if there was an increasing requirement of the above-mentioned inotropes with or without intra-aortic balloon counterpulsation along with afterload reduction with sodium nitroprusside. Patients who received less than 4 µg.kg-1.min-1) dopamine to increase renal perfusion were not considered to have low output syndrome. Patients who received vasoconstricting medications because of high cardiac output (>2.5 L.min-1.m-2) and low systemic vascular resistance were also not considered to have low output syndrome.
Techniques of Computerized-tomographic Angiography (Electronics)
All scans were performed on a third generation, 384 (2 x 192) slice, dual source CT scanner (SOMATOM FORCE, Siemens, Germany). It has a rotation time of up to 0.25 seconds with a temporal resolution of up to 66 milliseconds and spatial resolution of 0.24 mm.
No form of heart rate control was required. Retrospective ECG-gated CT angiography examination was performed after injection of non-ionic iodinated contrast (1.0 mL/Kg body weight) was administered via peripheral intravenous line at flow rate of 4.0 mL/s followed by a saline chaser (50 mL) injected at the same flow rate. A ‘bolus tracking’ method was used whereby CT acquisition was automatically triggered when contrast opacification threshold of 100 Hounsfield units (HU) was achieved in the ascending aorta on the monitoring sequence. Automated tube voltage selection and automated tube current modulation based on body habitus (CARE kV and CARE Dose4D, Siemens Healthcare) were enabled.
Slices were reconstructed of 0.6-mm section thickness and increment of 0.4 mm, using a medium sharp kernel (Bv40), with a model based iterative reconstruction strength level 3 (ADMIRE; Siemens Healthcare). Multiplanar reformatted images and volume rendered images were reconstructed and analysed.
